[ad_1]
Under the leadership of PD Dr. Niklas Klümper, Assistant Physician at the Department of Urology at the University Hospital Bonn (UKB) and Clinician Scientist of the BMBF-funded ACCENT program and PD Dr. Markus Eckstein, senior physician at the Institute of Pathology at the Uniklinikum Erlangen of the Friedrich-Alexander-University Erlangen-Nürnberg (FAU), an interdisciplinary research team has published new findings that indicate which patients with advanced urothelial carcinoma could benefit in particular from the new targeted therapy with the antibody-drug conjugate enfortumab vedotin. The study, published yesterday in the prestigious Journal of Clinical Oncology, identifies NECTIN4 amplification as a promising genomic biomarker for predicting treatment response to enfortumab vedotin. These findings could represent a significant advance in the improved treatment of this cancer.
As an alternative to chemotherapies used to treat aggressive advanced and metastatic urothelial carcinoma, a new class of drugs, known as antibody-drug conjugates, has recently become available. Enfortumab vedotin (EV) is the first drug in this new class to be approved by the EMA at for the treatment of patients and patients with metastatic urothelial carcinoma. Given the extremely promising results of the EV-302 trial, which showed a near doubling of survival with the combination therapy of EV and pembrolizumab, an immune checkpoint inhibitor, compared to conventional platinum-based chemotherapy in untreated patients with metastatic urothelial carcinoma, it is expected that the use of EV will increase significantly in the future.
Modern targeted oncology therapy
Antibody-drug conjugates consist of an antibody directed against a target structure on tumor cells and combined with a highly toxic chemotherapeutic agent. This combines the selectivity of targeted antibody therapy with the cytotoxic potential of conventional chemotherapy, which represents an innovative and new oncological therapeutic approach.
Research for more targeted therapy: precision oncology
Long-term efficacy of the new drug EV has so far been shown in an uncharacterized group of patients with metastatic urothelial carcinoma. The research team led by PD Dr. Niklas Klümper (UKB) and PD Dr. Markus Eckstein (Uniklinikum Erlangen) therefore wanted to analyze in more detail which patients benefit effectively from EV therapy in order to be able to use it in a more targeted manner – and conversely to identify patients who benefit less or not from EV, as they could possibly be treated more effectively with other therapies.
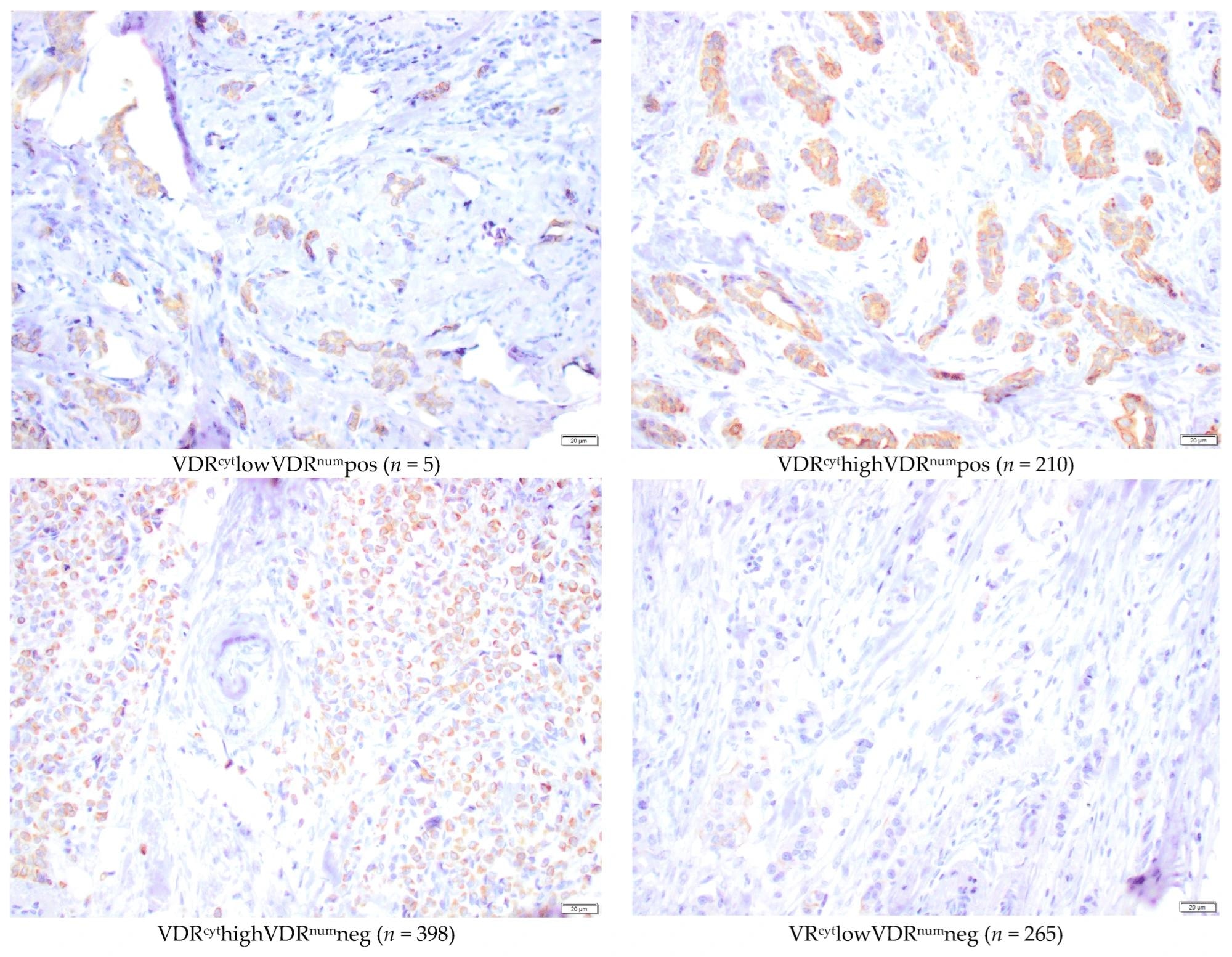
Nectin-4, the target structure of EV, is localized on chromosome 1q23.3. This gene segment is increased in about 20-25 percent of urothelial carcinomas, which is referred to as amplification. The new study aimed to investigate NECTIN4 amplifications as a potential genomic biomarker to predict treatment response to the drug EV in patients with advanced urothelial carcinoma.
“We have successfully developed and applied a simple FISH test (fluorescence in situ hybridization) that is specific for NECTIN4. This test proved to be a reliable method for identifying NECTIN4 amplification. Our studies showed that the presence of NECTIN4 amplification is a robust biomarker for response to EV therapy. In fact, over 90 percent of patients with NECTIN4 amplification showed tumor response to EV therapy, compared to about 30 percent of patients without this amplification”, says PD Dr. Markus Eckstein. These new findings can help to better select patients for this promising therapy in the future. “NECTIN4 amplification is a promising biomarker for predicting treatment response to EV. Excitingly, NECTIN4 amplification is also common in other solid tumors besides urothelial carcinoma, e.g. lung and breast cancer. The consideration of NECTIN4 amplification could therefore also be an exciting option for other tumor types in order to select patients for anti-NECTIN4-directed therapy in a more targeted manner. Further studies on this topic are needed, but our work could be the starting signal for the establishment of new targeted treatment strategies “, says PD Dr. Niklas Klümper.
Both study PIs also agree that without the great support of all the colleagues involved from the numerous oncology centers in Germany, Austria, the Netherlands and the USA, and of course without the patients’ consent to participate in the study, it could never have been carried out successfully.
The study was funded and initiated by the German Research Foundation (DFG) as part of the DFG Young Investigator Academy UroAgeCare of the German Society of Urology (DGU). “This underlines the high relevance of funding clinician scientist programs for medical progress,” says Prof. Michael Hölzel, mentor of PD Dr. Klümper within the program.
Source:
Journal reference:
Klümper, N., et al. (2024) NECTIN4 Amplification Is Frequent in Solid Tumors and Predicts Enfortumab Vedotin Response in Metastatic Urothelial Cancer. Journal of Clinical Oncology. doi.org/10.1200/JCO.23.01983.
[ad_2]
Source link