[ad_1]

Researchers at KIST have developed a fiber-based adsorbent capable of recovering gold from electronic waste with over 99.9% efficiency. Credit: SciTechDaily.com
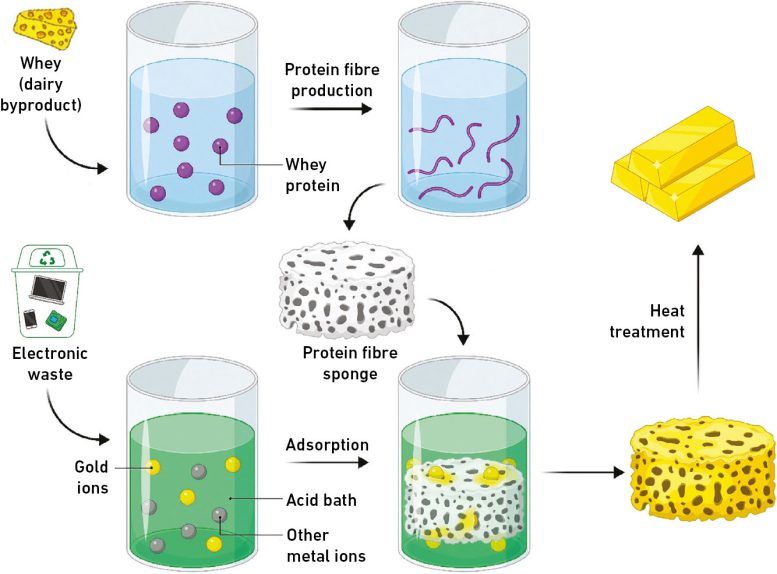
A fibrous adsorbent selectively recovers high-purity gold from waste. Dramatically reduces the cost and time of the recovery process and enables material to be mass-produced and repeatedly recycled.
Korea relies on imports for most of its metal resources, and in recent years, due to resource depletion and rising raw material prices, ‘circular resources’ that recycle waste metal resources have emerged.
In response, SK hynix has established a mid- to long-term plan to increase the percentage of copper, gold, etc. recovered and reused from waste generated in the semiconductor manufacturing process to more than 30% by 2030, and Samsung Electronics is running a collection program for used mobile phones in cooperation with E-circulation Governance, a non-profit corporation. The global circular economy market is expected to more than double in size from approximately $338 billion in 2022 to approximately $712 billion in 2026.
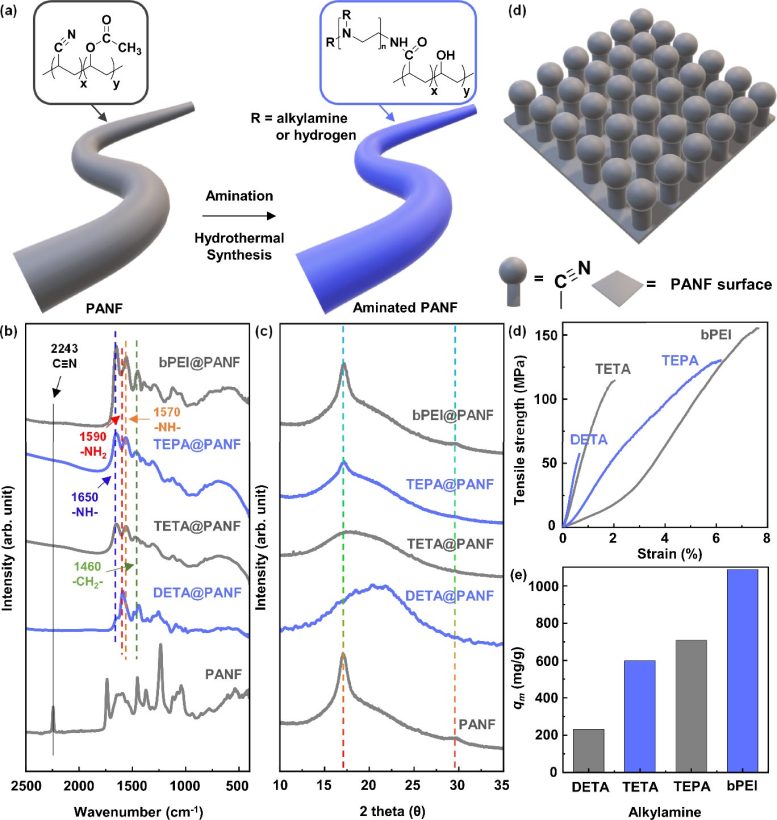
Figure 1. Preparation and physicochemical characteristics of the aminated polyacrylonitrile fibers (PANFs). Representative illustrations of PANF (a) before and after coupling reaction of various alkylamine molecules. Different colors of PANF and amine-laden polymeric fiber (ALPF) represent different functional groups of nitrile and alkylamines. (b) FT-IR spectra of PANF before and after coupling reaction of different alkylamines using diethylenetriamine (DETA), triethylenetetramine (TETA), tetraethylenepentamine (TEPA), and branched poly(ethyleneimine) (bPEI). (c) XRD patterns of the PANF and aminated PANFs. (d) Stress-strain curves of the PANF and aminated PANFs. (e) Maximum adsorption capacity (qm) of the aminated PANFs for Au(III) ions. Initial concentration (Ci) and pH value of the Au solution were 1000 mg/L and 1, respectively. Credit: Korea Institute of Science and Technology
Breakthrough in Metal Recovery Technology
In this context, a team led by Dr. Jae-Woo Choi of the Water Resource Cycle Research Center at the Korea Institute of Science and Technology (KIST) announced that they have developed a technology that can selectively recover high-purity gold from electrical and electronic waste containing various metals using textile materials.
Adsorbents for metal recovery are generally granular in shape to increase adsorption efficiency based on high specific surface area, but they are difficult to control underwater, resulting in low recovery rates and even secondary environmental pollution. On the other hand, fiber-like materials are easy to control underwater and can be made into various shapes through the weaving process, so they have high potential for industrial application. However, due to their thin thickness and low strength, they are easily broken when gold recovery is applied to the support.
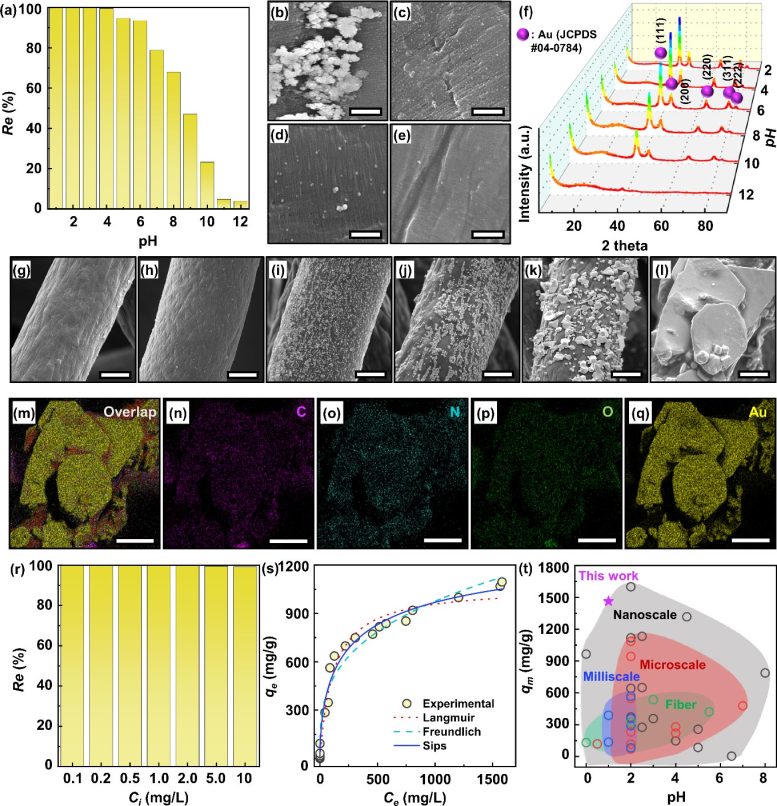
Figure 2. Au recovery performance of the ALPF. (a) Effect of pH in Au solution on the Au recovery performance of the ALPF. Ci, t, and adsorbent doses were 100 mg/L, 24 h, and 0.5 g/L, respectively. FESEM images of the ALPF surface after Au recovery at pH of (b) 3, (c) 6, (d) 9, and (e) 12, showing the Au(0) particles on the ALPF surface. Scale bar is 1 μm. (f) XRD patterns of the ALPF after Au recovery in the pH range of 2-12. (g-l) FESEM images of the ALPF after Au recovery at Ci of (g) 0.1, (h) 1, (i) 10, (j) 100, (k) 500, and (l) 1000 mg/L for 24 h with stirring at 200 rpm. pH was adjusted to 1. Scale bar is 20 μm. (m-q) FESEM-EDS mapping of the chemical elements distributions for the ALPF after Au recovery at Ci of 1000 mg/L: (m) overlap, (n) carbon, (o) nitrogen, (p) oxygen, and (q) gold. Scale bar is 20 μm. (r) Recovery efficiencies of the ALPF for Au recovery in a low Ci range of 0.1-100 mg/L. (s) Adsorption isotherm test result of the ALPF adsorbent. The experimentally obtained data were fitted by three representative isotherm equations of Langmuir, Freundlich, and Sips models. (t) Comparison of qm and optimum pH value for the ALPF adsorbent with those of the adsorbents best reported in the literature, classified by the adsorbent size (nano, micro, and milliscale) or shape (particle and fiber). Credit: Korea Institute of Science and Technology
Enhanced Gold Recovery Techniques
KIST researchers have chemically immobilized alkaline molecules on the surface of polyacrylonitrile (PANF) fibers to improve both molecular gold recovery performance and structural stability. The amine-containing polymer fiber has a dramatically larger surface area, which can improve the adsorption performance of gold ions (Au) in waste by up to 2.5 times (from 576 mg/g to 1,462 mg/g) compared to the team’s previously developed granular gold adsorption material.
The developed fibrous adsorbent not only showed a gold recovery efficiency of more than 99.9% in solutions obtained by leaching real CPUs, but also achieved a gold recovery efficiency close to 100% in a wide range of pH 1-4, which includes most waste liquids. It is particularly noteworthy that only gold ions can be recovered with a high purity of over 99.9%, even in the presence of 14 other metal ions coexisting in the solution. Furthermore, the gold recovery rate was maintained at 91% even after 10 uses, demonstrating excellent reusability.
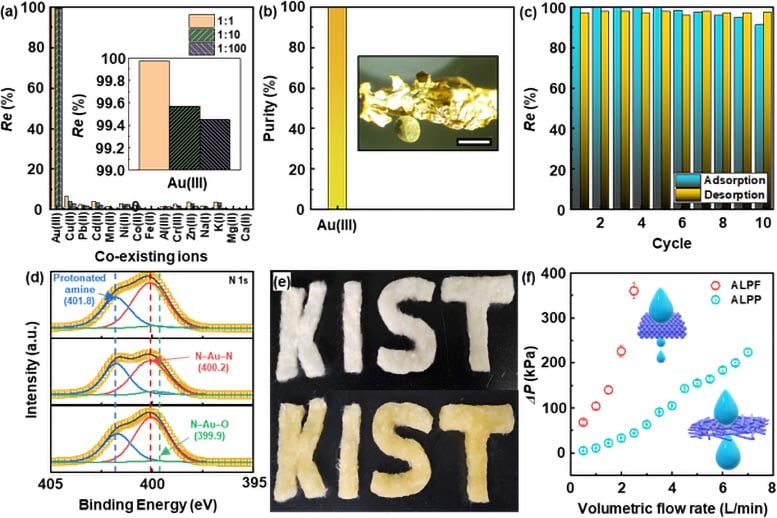
Figure 3. Applicability of the ALPF adsorbent for Au recovery processes. (a) Adsorption selectivity of the ALPF for Au(III) in the presence of coexisting metal ions including Cu(II), Pb(II), Cd(II), Mn(II), Ni(II), Co(II), Fe(II), Al(III), Cr(III), Zn(II), Na(I), K(I), Mg(II), and Ca(II). Ci of Au(III) was set to 10 mg/L, and those of other metal ions were set to 10, 100, and 1000 mg/L. Solution pH was adjusted to 1. (b) Purity of the recovered Au(0) by the ALPF. Inset shows an optical microscope image of the recovered Au(0). (c) Repetitive adsorption/desorption test of Au(III) using the ALPF adsorbent. Ci of Au(III) was set to 10 mg/L. Adsorption process was conducted for 24 h. Solution pH was adjusted to 1. Desorption process was conducted for 24 h using 0.5 acidic thiourea solution in 1.0 M HCl. (d) Deconvoluted HRXPS spectra of the ALPF for N 1s, showing the chemical change for amine groups of the ALPF adsorbent during the repetitive adsorption-desorption cycles. (e) Photograph of felts consisting of PANF (top) and ALPF (bottom), indicating that the fibrous adsorbent can be transformed into a desired shape. Scale bar is 2 cm. (f) Effect of the adsorbent shapes on the pressure drop in a column filled with the adsorbents. Each pressure drop in the column filled with the adsorbents was measured according to the volumetric flow rate. Credit: Korea Institute of Science and Technology
Conclusion and Future Prospects
“By enabling efficient and eco-friendly metal resource recovery, the fiber-type adsorbent developed by KIST can reduce Korea’s dependence on resource imports and prepare for the risk of rising raw material prices,” said Dr. Jae-Woo Choi. “We plan to expand the scope of future research to selectively recover various target metals in addition to gold, said Dr. Youngkyun Jung.”
Reference: “Efficient and selective gold recovery using amine-laden polymeric fibers synthesized by a steric hindrance strategy” by Youngkyun Jung, Su-Jin Yoon, Kyung-Won Jung and Jae-Woo Choi, 12 February 2024, Chemical Engineering Journal.
DOI: 10.1016/j.cej.2024.149602
[ad_2]
Source link