[ad_1]
Scientists at the Karolinska Institute in Sweden have conducted a systematic review and meta-analysis to determine the relationship between dietary antioxidant intake and type 2 diabetes risk.
The study is published in the journal Advances in Nutrition.
 Review: Vitamins C, E, and beta-carotene and risk of type 2 diabetes: a systematic review and meta-analysis. Image Credit: alicja neumiler / Shutterstock
Review: Vitamins C, E, and beta-carotene and risk of type 2 diabetes: a systematic review and meta-analysis. Image Credit: alicja neumiler / Shutterstock
Background
Type 2 diabetes is a serious metabolic disease characterized by reduced secretion or impaired functioning of insulin and subsequent increase in blood glucose level (hyperglycemia). Insulin resistance and pancreatic beta-cell dysfunction are two major hallmarks of the disease.
More than 10% of the global population is currently affected by type 2 diabetes. Its prevalence is sharply increasing worldwide mainly because of an increasing inclination towards unhealthy food habits and sedentary lifestyles.
Diet plays an important role in regulating the body’s metabolism, among various lifestyle factors. Evidence indicates that an increased adherence to healthy diets, such as the Mediterranean or DASH (Dietary Approach to Stop Hypertension), can significantly reduce the risk of type 2 diabetes. The fundamental characteristic of these diets is a higher intake of plant-based foods rich in antioxidants, including vitamins C, E, and beta-carotene.
In this systematic review and meta-analysis, scientists have assessed whether consumption of dietary vitamins C, E and beta-carotene can reduce the risk of type 2 diabetes.
Study design
The scientists searched various electronic databases to identify studies investigating the association between dietary intakes, circulating levels, or supplementation of vitamin C, E, and beta-carotene and type 2 diabetes incidence or insulin resistance/sensitivity and beta cell function in non-diabetic individuals.
The final screening led to the identification of 25 prospective observational studies and 15 randomized controlled trials. Moderate and serious risks of bias were observed in 21 and 4 observational studies, respectively. Among randomized controlled trials, 13 had a low risk of bias, and 2 had some concerns.
Important observations
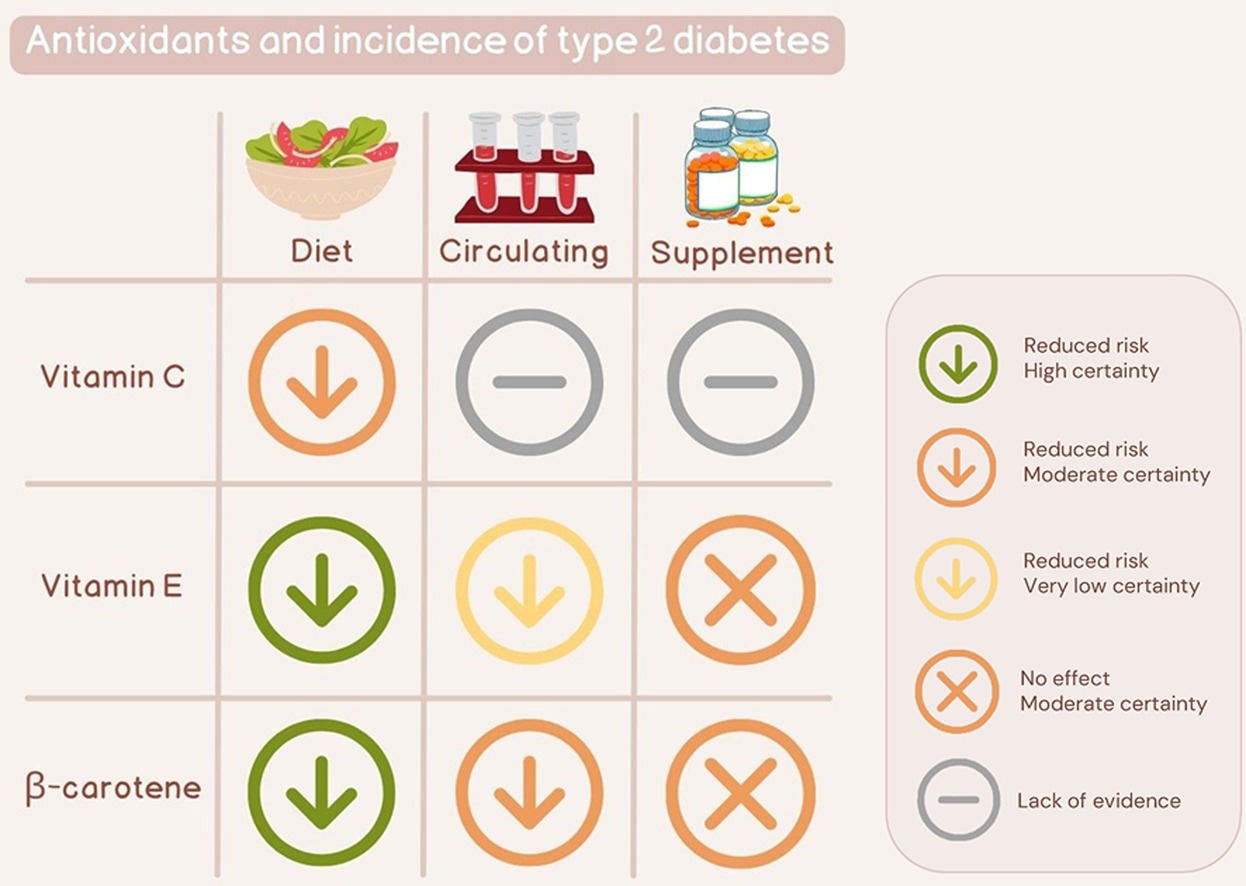
The study found that moderate intakes of vitamins C, E, and beta-carotene can reduce the risk of type 2 diabetes.
Vitamin C
The meta-analysis of observational studies revealed that a vitamin C intake of up to 70 mg per day can reduce type 2 diabetes risk by 24%. However, no further risk reduction was observed for an intake higher than this level.
An inverse association was observed between dietary intake of vitamin C and insulin resistance. Vitamin C intake also showed a positive impact on beta cell function.
Vitamin E
The meta-analysis of observational studies revealed that the vitamin E intake of up to 12 mg per day can reduce the risk of type 2 diabetes by 28%. Similar to vitamin C, no further risk reduction was observed for an intake higher than this level.
The meta-analysis of randomized clinical trials revealed that vitamin E supplementation does not have any protective effect against type 2 diabetes development. However, vitamin E supplementation showed a positive impact on insulin sensitivity.
Beta-carotene
The meta-analysis of observational studies revealed that the beta-carotene intake of up to 4 mg per day can reduce the risk of type 2 diabetes by 22%. No further risk reduction was observed above this level.
The meta-analysis of randomized controlled trials revealed that beta-carotene supplementation cannot reduce the risk of type 2 diabetes. It was also observed that circulating beta-carotene can reduce insulin resistance and increase insulin sensitivity.
Study significance
This systematic review and meta-analysis found an inverse association between dietary and circulating vitamins C, E, and beta-carotene and the risk of type 2 diabetes. However, no protective efficacy of supplementation with these antioxidants has been observed against type 2 diabetes.
The robust antioxidant properties of these vitamins are mainly responsible for their anti-diabetic effects. Vitamin C is a water-soluble vitamin commonly found in fruits and vegetables. It can remove free radicals in the body’s hydrophilic compartments and regenerate vitamin E from its oxidized form.
Vitamin E is a fat-soluble vitamin commonly found in nuts, seeds, and vegetable oils. It can prevent lipid peroxidation and protect lipid parts of the body, such as cell membranes. Beta-carotene is a fat-soluble provitamin A carotenoid commonly found in fruits and vegetables. Similar to vitamin E, beta-carotene can protect lipid parts of the body from free radical-mediated damage.
Article Revisions
- Mar 27 2024 – Addition of Graphical abstract illustration.
[ad_2]
Source link