[ad_1]
Frailty, a clinical syndrome affecting almost 10% of the elderly, undermines their ability to handle stress.
Nutrition plays a key role in frailty development, with protein supplements often used to boost strength and physical function in frail older adults. Although meat is a rich protein source, its impact on the health of frail individuals remains under-researched.
A recent study in The Journal of Nutrition, Health and Aging explores how meat consumption influences the risk of death in frail populations.
 Study: Associations between meat consumption and all-cause and cause-specific mortality in middle-aged and older adults with frailty. Image Credit: KucherAV/Shutterstock.com
Study: Associations between meat consumption and all-cause and cause-specific mortality in middle-aged and older adults with frailty. Image Credit: KucherAV/Shutterstock.com
About frailty
Frailty arises from diminished physiological capacity due to existing deficiencies across various bodily systems, leading to increased vulnerability to stressors and a higher demand for medical care in the elderly.
Recent studies aim to enhance the well-being of frail individuals by investigating the roots of frailty and tracking the progression of this condition.
The role of meat in frailty
Meat, as a food category, includes red meat, fish, and poultry. Rich in proteins and micronutrients, meat is important for building and maintaining muscle strength, enhancing physical strength, and minimizing the incidence of malnutrition in the elderly.
Even though frail people need to eat more protein, prior research has demonstrated a positive association of red meat with frailty, unlike other types of protein.
This could be due to the high saturated fat content in red meat that is associated with increased cardiovascular disease (CVD) risk.
In addition, processed meat contains nitrites and other preservatives that may trigger oxidative stress and inflammation, both of which cause CVD and metabolic disease.
Such findings have led to recommendations, both by the World Health Organization (WHO) and many national nutritionist bodies, to eat less red meat and processed meat.
What did the study show?
There were around 20,000 participants, with a mean age of 58 years. About 60% were female. About 38% and 12% ate red meat and processed meat, respectively.
About a fifth consumed poultry. Increased frequency of poultry consumption was associated with a lower risk of death from all causes. Compared to those who had poultry less than once a week, those who had it 1-2 times had a 10% lower risk, while those who ate it >4 times a week had a 33% lower risk.
Deaths from cancer were also lower, at 10% and 20%, for those who had poultry 1-2 times vs >4 times a week. Moreover, mortality from CVD was reduced by 15%, 25%, and 50%, among those who ate it 1-2, 2-4, and >4 times a week, respectively.
The opposite trends were seen among those who had higher processed meat consumption. All-cause mortality was increased by 10% and 20% among those who ate processed meat 2-4 vs >4 times a week, respectively.
Deaths from CVD were higher by 15% and 25% among those who had processed meat 1-4 times vs >4 times a week, respectively.
There was a U-shaped relationship between red meat intake and mortality. The death rates from all causes, cancer, and CVD were all lower among those who ate red meat up to 2 times a week, but only the first was significant.
The risk of all-cause deaths was 14% lower in this group compared to those who had red meat less than once a week.
For each additional 25 g of red meat, the risk of all-cause mortality and deaths from CVD among the frail increased by 7% and 16%, respectively. This was not the case for overall meat consumption.
If processed meat was replaced by oily fish, like sardines, the risk of all-cause death was 5% lower. If substituted by poultry, it was 9% lower for all-cause deaths and 7% less for cancer deaths. CVD mortality was reduced by 13%.
If fish replaced red meat, all-cause mortality went down by 3%, but if oily fish was consumed, it went down by 6%. Substitution with cheese led to a 4% reduction but by 10% for poultry.
These substitutions were also more heart-friendly, with poultry being associated with 13% lower CVD mortality vs 5% for cheese and for fish.
Interestingly, increased red meat consumption was linked to a higher risk of all-cause mortality among males only.
This risk was also stronger for processed meat consumption among males and those with a body mass index (BMI) of 25 kg/m3 or higher. BMI was also similarly linked to increased risk for all-cause mortality with increasing meat consumption.
Lessons to learn
“We need much more research on optimal dietary components for people with frailty because current clinical guidelines are mainly based on expert consensus because of the lack of an evidence base.”
These findings from a study that explores the different types of meat consumption in relation to mortality risk in frail individuals supply a significant foundation for such recommendations.
They corroborate earlier studies showing the adverse impact of processed meat on all-cause and cardiovascular mortality, which outweighs the potential benefit in terms of the protein supplied.
The association of red meat intake >2 times a week with increased mortality has not been reported by other studies but may be due to the saturated fat content, which may boost the risk of CVD.
The results indicate that “poultry and fish could be a healthier alternative to red and processed meat among frail individuals.”
Further research is necessary to explore the interaction of BMI with processed meat consumption and mortality risk. The reasons for increased mortality with red meat intake in males remain unexplained, inviting future studies.
Journal reference:
- Jie Chen a, Weihao Xu, Lintao Dan, Junhan Tang b, Jirong Yue e, Emiel O. Hoogendijk f, and Chenkai Wu (2024). Associations between meat consumption and all-cause and cause-specific mortality in middle-aged and older adults with frailty. The Journal of Nutrition, Health and Aging. doi: http://dx.doi.org/10.1016/j.jnha.2024.100191.
[ad_2]
Source link












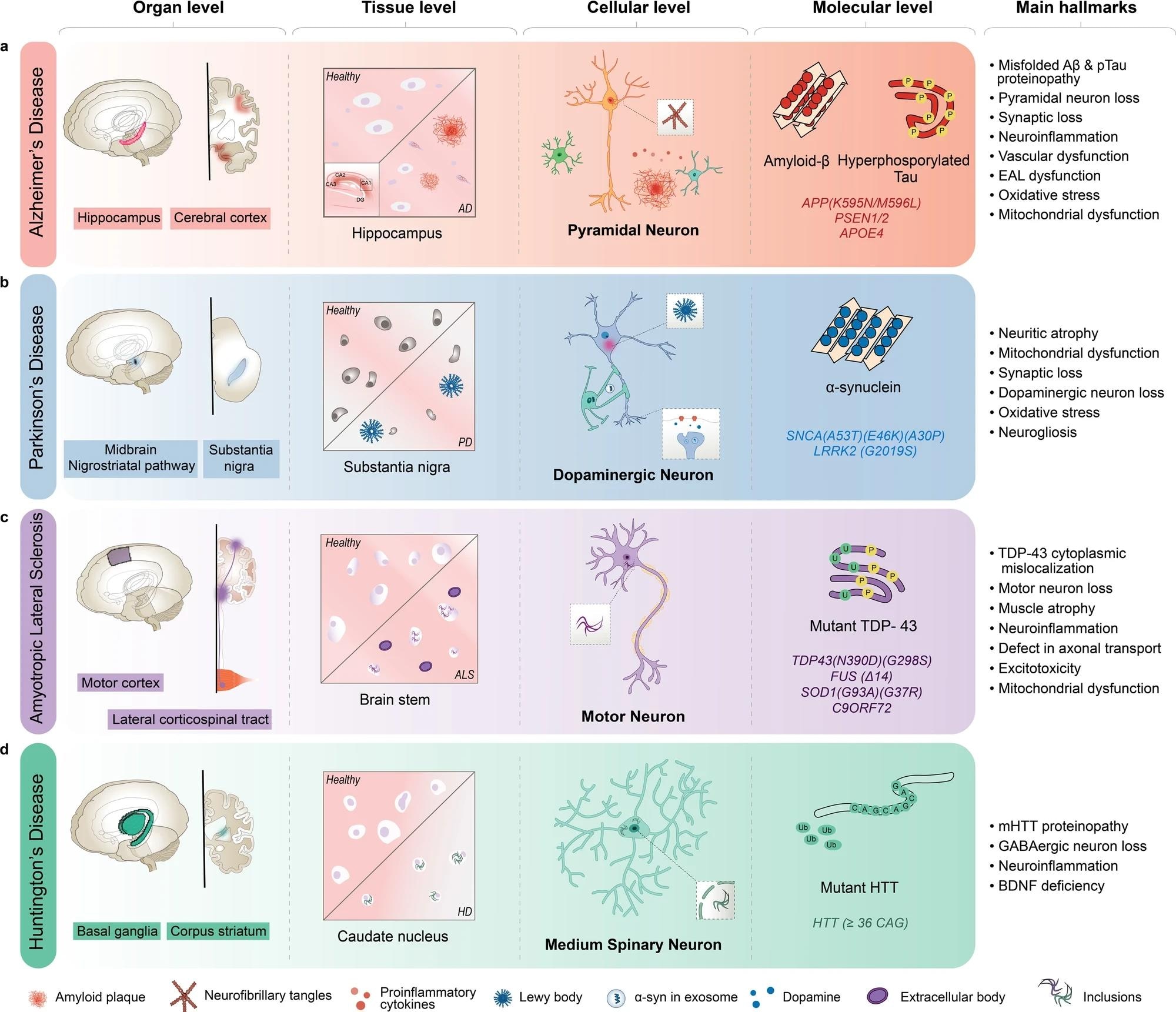 AD is characterized by the inclusion of misfolded amyloid-β (Aβ) and neurofibrillary tangles in pyramidal neurons, primarily in the hippocampus and cortex regions of the brain. b PD is characterized by Lewy body aggregates composed of misfolded α-synuclein and degeneration of dopaminergic neurons in the substantia nigra region of the brain. c ALS is characterized by including mutant TAR DNA-binding protein 43 (TDP-43) and other proteins, degeneration of motor neurons in the motor cortex and spinal cord, and muscle atrophy with dysfunctional proteins. d HD is characterized by including mutant Huntingtin protein (mHTT) and degeneration of medium spiny neurons in the basal ganglia, and corpus striatum of the brain. AD Alzheimer’s disease, ALS amyotrophic lateral sclerosis, BDNF brain-derived neurotrophic factor, EAL endosomal-autophagic-lysosomal pathway, GABA gamma-aminobutyric acid, HD Huntington’s disease, PSEN presenilin 1, SNCA synuclein alpha.
AD is characterized by the inclusion of misfolded amyloid-β (Aβ) and neurofibrillary tangles in pyramidal neurons, primarily in the hippocampus and cortex regions of the brain. b PD is characterized by Lewy body aggregates composed of misfolded α-synuclein and degeneration of dopaminergic neurons in the substantia nigra region of the brain. c ALS is characterized by including mutant TAR DNA-binding protein 43 (TDP-43) and other proteins, degeneration of motor neurons in the motor cortex and spinal cord, and muscle atrophy with dysfunctional proteins. d HD is characterized by including mutant Huntingtin protein (mHTT) and degeneration of medium spiny neurons in the basal ganglia, and corpus striatum of the brain. AD Alzheimer’s disease, ALS amyotrophic lateral sclerosis, BDNF brain-derived neurotrophic factor, EAL endosomal-autophagic-lysosomal pathway, GABA gamma-aminobutyric acid, HD Huntington’s disease, PSEN presenilin 1, SNCA synuclein alpha.