[ad_1]
In a recent review article published in the journal Antioxidants, researchers explored how environmental factors impact the brain development of fetuses and neonates, emphasizing inflammation and oxidation stress as common denominators across various stressors.
Their conclusions highlight the critical role of healthy intrauterine environments for promoting fetal brain development and stress the importance of interventions that aim to reduce modifiable stress factors during pregnancy.
 Review: Impact of Maternal Environment and Inflammation on Fetal Neurodevelopment. Image Credit: Sebastian Kaulitzki / Shutterstock
Review: Impact of Maternal Environment and Inflammation on Fetal Neurodevelopment. Image Credit: Sebastian Kaulitzki / Shutterstock
Background
The development of the human brain, which commences during the second to third week of gestation and continues through childhood, is influenced by a combination of genetic, epigenetic, and environmental factors.
Key developmental milestones occur during specific gestational periods, such as cellular migration in the neocortex and extensive neurogenesis between the eighth and eighteenth weeks.
Maternal environmental exposures during the prenatal and antenatal periods can impact intrauterine development and the short—and long-term health of the offspring, potentially raising the risks of developing non-communicable diseases in adulthood.
These exposures can epigenetically modify placental and fetal phenotypes, affecting organ structure, metabolism, and physiology. However, understanding the precise molecular mechanisms linking external factors to neurodevelopmental alterations remains challenging.
In this review, researchers aimed to explore the effects of various maternal environmental exposures, including nutrition, lifestyle, stress, and pollution, on fetal brain development and neonatal neurodevelopment-related outcomes, drawing from a comprehensive literature search that encompassed human and animal studies published within the last 15 years.
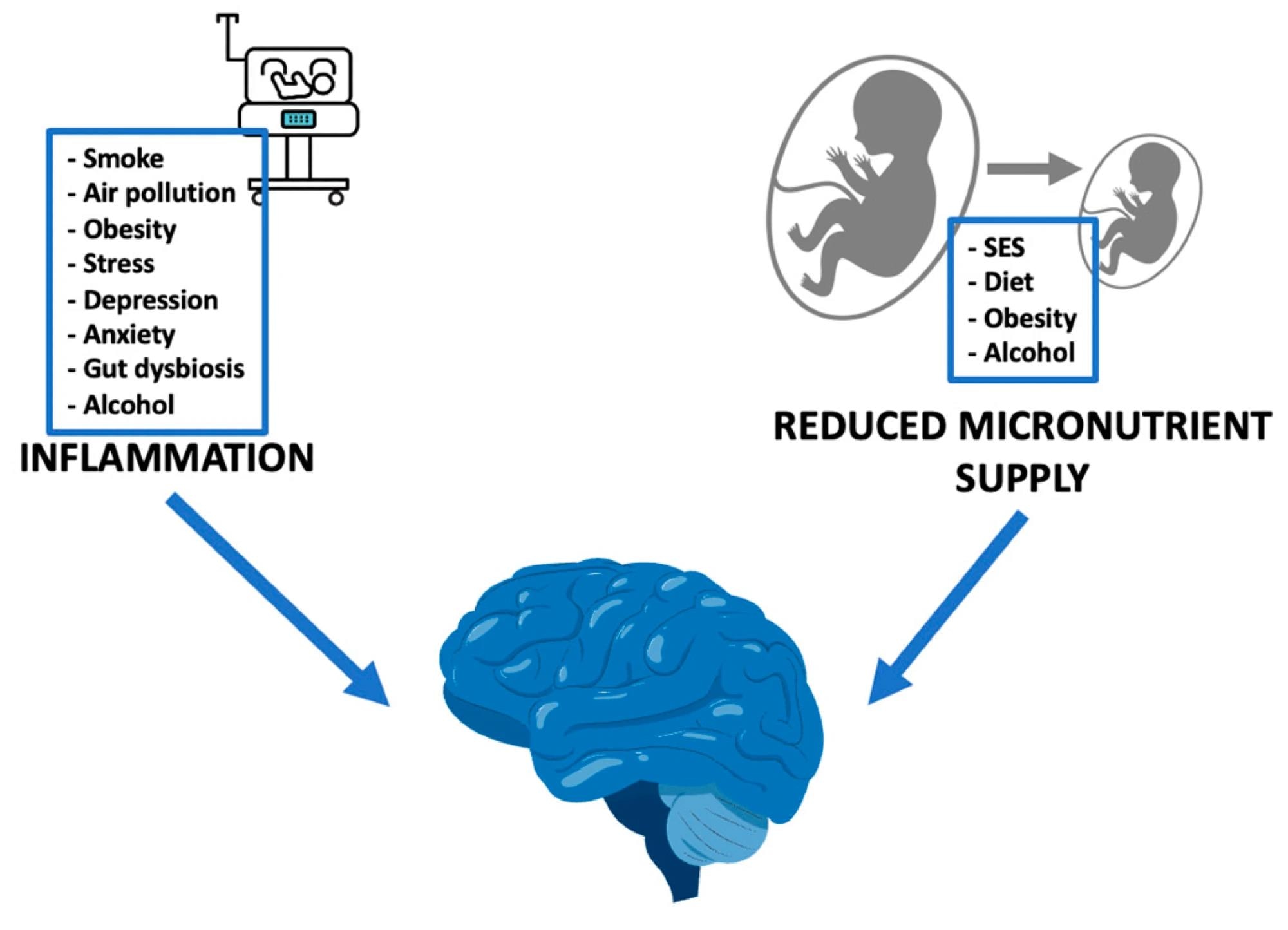 External stimuli, through inflammation and reduced micronutrient supply, impact on fetal neurodevelopment. SES: socioeconomic status.
External stimuli, through inflammation and reduced micronutrient supply, impact on fetal neurodevelopment. SES: socioeconomic status.
Maternal environmental exposures
Maternal nutrition plays a crucial role in fetal neurodevelopment, with evidence suggesting that both insufficient and unhealthy dietary patterns during pregnancy can affect fetal brain development.
For example, the Mediterranean diet, characterized by nutrient-rich foods, has been associated with positive neurodevelopmental outcomes in offspring.
However, deficiencies in micronutrients like docosahexaenoic acid (DHA), folate, and iodine and excessive intake of macronutrients have been linked to adverse neurodevelopmental effects.
The Western diet, in particular, is known to be high in macronutrients but poor in micronutrients. Affordable but low-quality foods characterize it. Mouse models suggest that pow-protein diets may also be associated with neurodevelopmental delays.
Alterations in maternal gut microbiota during pregnancy have been linked to worse behavioral outcomes in offspring, potentially mediated through inflammation and metabolic endotoxemia.
Maternal obesity and a high-fat diet have also been implicated, with animal models and epidemiological studies suggesting associations with cognitive deficits, attention deficit hyperactivity disorder (ADHD), autism, and psychoses in offspring.
Additionally, maternal depression, anxiety, and stress can lead to disturbed fetal neurodevelopment, potentially resulting in altered brain structure and function, as evidenced by investigations using human and animal models.
Smoking and alcohol consumption during pregnancy have well-documented detrimental effects on fetal neurodevelopment, including increased risks of ADHD, autism, schizophrenia, and behavioral issues.
Exposure to air pollution, mainly particulate matter, and polycyclic aromatic hydrocarbons, has been linked to neurodevelopmental disorders in offspring, with oxidative stress and inflammatory responses implicated as underlying mechanisms.
Socioeconomic status also plays a role, with disadvantaged conditions associated with adverse pregnancy outcomes and poorer neurodevelopmental outcomes in children.
Underlying mechanisms
Numerous studies have linked pathological pregnancy conditions like fetal growth restriction (FGR) and preterm birth (PTB) with neurodevelopmental issues in offspring. Two main mechanisms are implicated: altered fetal nutrient supply and intrauterine inflammation involving placental factors.
FGR, often due to placental dysfunction, leads to chronic fetal hypoxia and undernutrition, impacting fetal brain development despite adaptive responses like brain sparing.
PTB, often linked to maternal inflammation or infection, presents various neurodevelopmental challenges, including disruptions in axonal and neuronal development and brain abnormalities like cerebral palsy.
Inflammation during pregnancy, exacerbated by factors like obesity, stress, and environmental pollutants, influences insulin and neurotransmitter signaling, affecting synaptic plasticity and neurotrophic factor expression.
The activation of maternal immune systems contributes to mitochondrial dysfunction and oxidative stress, perpetuating a cycle of oxidative stress and inflammation that disrupts fetal brain development.
These conditions and exposure to chemicals can compromise the blood-brain barrier and lead to additional impairments in fetal brain development.
Overall, maternal health conditions and external factors collectively contribute to increased maternal inflammation, which impacts fetal neurodevelopment and potentially leads to long-term neurological consequences in offspring.
Conclusions
The review emphasizes how external factors during pregnancy affect fetal growth and brain development, impacting long-term neurodevelopment.
Conditions like PTB and FGR alter brain morphometry, often due to inflammation and changes in nutrient supply. Evidence suggests they are influenced by maternal health and environmental factors like air pollution and stress.
Understanding and addressing these modifiable risk factors is crucial for improving both individual and public health outcomes. This highlights the importance of preventive measures and further longitudinal research.
Journal reference:
- Impacts of maternal environment and inflammation on fetal neurodevelopment. Lubrano, C., Parisi, F., Cetin, I. Antioxidants (2024). DOI: 10.3390/antiox13040453, https://www.mdpi.com/2076-3921/13/4/453
[ad_2]
Source link