[ad_1]
The transition from the esophagus to the stomach is a delicate region from a medical point of view, often associated with pathological disorders leading to cancer. An international research team has now gained new insights into this region. These pave the way for new prevention and treatment options.
The meeting point of the stomach and esophagus, the so-called gastro-esophageal junction, is a region of the human body that is not well-suited to the modern lifestyle. Stress, alcohol, nicotine and severe obesity are often triggers for pathological changes to the mucosal membrane in this area, often resulting in esophageal cancer.
An international research team has now gained new insights into the development of the cells, their communication with each other, and their regulation at the junction of the esophagus and stomach. With the help of specially developed mini-organs, so-called organoids, and with techniques that make it possible to track and profile individual cells, they have been able to follow the development of the gastro-esophageal junction from embryonic to adult stage in detail using animal experiments.
New insights into the development of the gastrointestinal tract
Their results reveal the complex communication at the cellular level and the specific pathways that these cells use to communicate. They provide new insights into the development of the gastro-esophageal junction and thus have significant implications for the understanding, prevention and treatment of gastrointestinal diseases. At the same time, they present new starting points for medical research and the development of new therapies.
Cindrilla Chumduri is responsible for this study, which has now been published in the journal Nature Communications. Until recently, the infection and cancer biologist was a research group leader at the Department of Microbiology at Julius-Maximilians-Universität Würzburg (JMU); she is now an associate professor at Aarhus University (Denmark). Other participants came from Charité – Universitätsmedizin and the Max Planck Institute for Infection Biology in Berlin.
“This collaboration underlines the importance of different expertise to improve our understanding of the biology of the gastrointestinal tract,” says Chumduri.
She herself has many years of experience in research with organoids. Among other things, she has used mini-organs she developed to study how cells in the cervix degenerate and turn cancerous – another region where different types of mucosal cells collide.
Where different epithelia meet
The squamous epithelia of the esophagus and the columnar epithelia of the stomach meet at the gastroesophageal junction.”
Dr. Naveen Kumar Nirchal, one of the first authors of the study
The area is known as a “hotspot for the development of metaplasia” – the replacement of one type of cell by another.
Barrett’s esophagus, a precursor to esophageal cancer, often develops there, the number of cases of which has increased dramatically in the Western world over the past four decades. “Barrett’s esophagus is characterized by the replacement of the resident squamous epithelium of the esophagus by other cell types that are not normally found in this tissue,” says the scientist.
However, it is still unclear why this region is so susceptible to this process. In order to better understand this transformation, it is therefore first necessary to decipher the normal development process in detail – from embryo to mature adult. “This is the only way to determine the tissue changes that trigger the progression of the disease, explains Dr. Rajendra Kumar Gurumurthy, another researcher of the study.
A never-before-seen insight into the development of this region
This has now been achieved: By using a novel approach that combines organoid and mouse models with advanced single-cell transcriptome analyses over time and space, the research team has shed light on the complex developmental process of the gastroesophageal junction. “We were able to provide unprecedented insight into the development of this region from the embryonic stage to adulthood in mice and identify the intricate composition of the cells involved and how they develop,” explains Pon Ganish Prakash, another scientist involved in the study.
The work shows the sophisticated communication between different cell types within the gastroesophageal junction and the signaling pathways involved. “This understanding opens up new avenues for research into gastrointestinal diseases,” says Cindrilla Chumduri.
Above all, the precision of the single-cell analysis in their study opens new doors to understanding how pathological processes develop and to developing innovative treatments, the team writes in its study. The work will therefore be a “cornerstone for understanding the development of such diseases” and will significantly influence the approach to the early detection and treatment of diseases in this important part of the digestive system.
Source:
Journal reference:
Kumar, N., et al. (2024). Decoding spatiotemporal transcriptional dynamics and epithelial fibroblast crosstalk during gastroesophageal junction development through single cell analysis. Nature Communications. doi.org/10.1038/s41467-024-47173-z.
[ad_2]
Source link






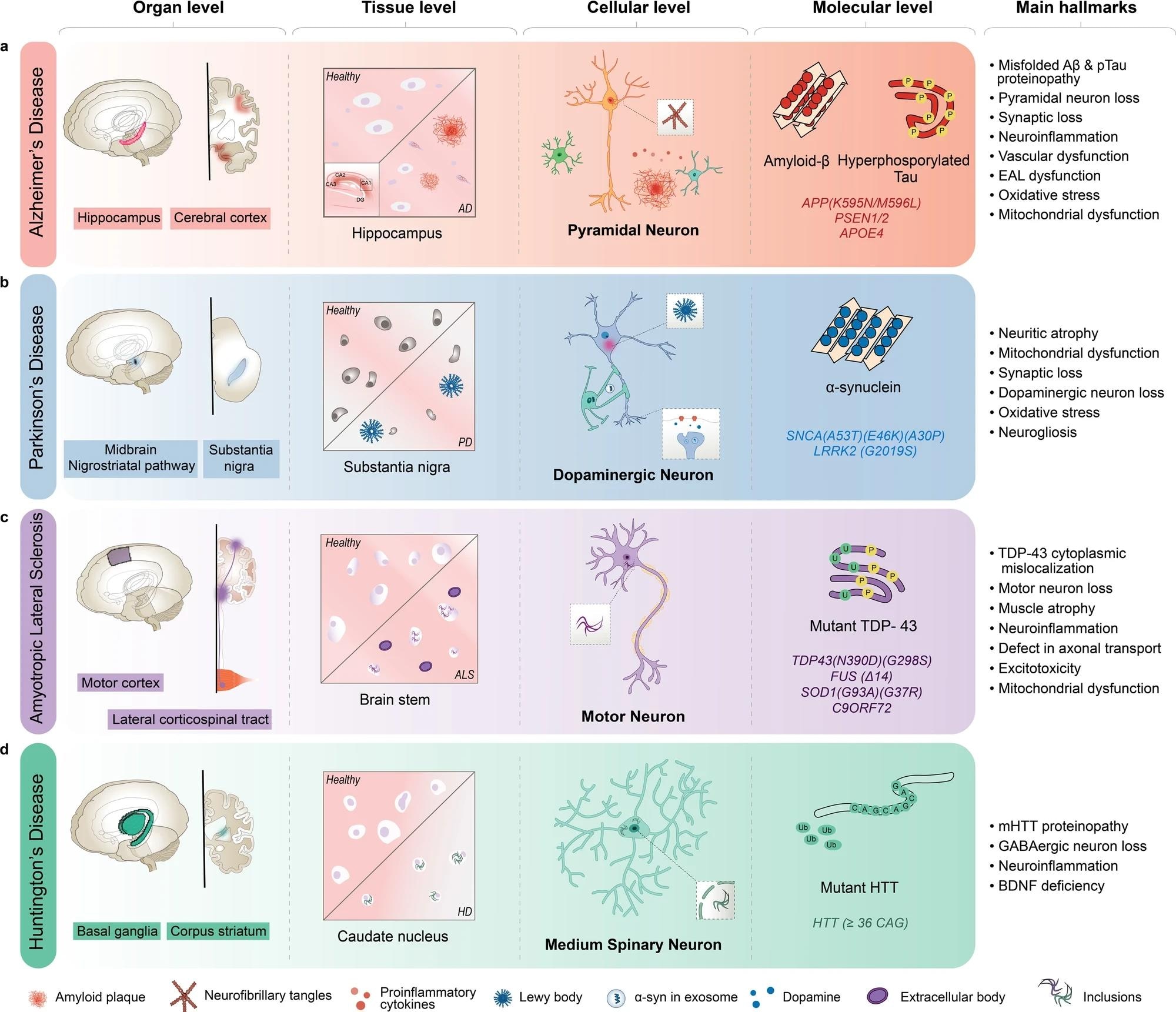 AD is characterized by the inclusion of misfolded amyloid-β (Aβ) and neurofibrillary tangles in pyramidal neurons, primarily in the hippocampus and cortex regions of the brain. b PD is characterized by Lewy body aggregates composed of misfolded α-synuclein and degeneration of dopaminergic neurons in the substantia nigra region of the brain. c ALS is characterized by including mutant TAR DNA-binding protein 43 (TDP-43) and other proteins, degeneration of motor neurons in the motor cortex and spinal cord, and muscle atrophy with dysfunctional proteins. d HD is characterized by including mutant Huntingtin protein (mHTT) and degeneration of medium spiny neurons in the basal ganglia, and corpus striatum of the brain. AD Alzheimer’s disease, ALS amyotrophic lateral sclerosis, BDNF brain-derived neurotrophic factor, EAL endosomal-autophagic-lysosomal pathway, GABA gamma-aminobutyric acid, HD Huntington’s disease, PSEN presenilin 1, SNCA synuclein alpha.
AD is characterized by the inclusion of misfolded amyloid-β (Aβ) and neurofibrillary tangles in pyramidal neurons, primarily in the hippocampus and cortex regions of the brain. b PD is characterized by Lewy body aggregates composed of misfolded α-synuclein and degeneration of dopaminergic neurons in the substantia nigra region of the brain. c ALS is characterized by including mutant TAR DNA-binding protein 43 (TDP-43) and other proteins, degeneration of motor neurons in the motor cortex and spinal cord, and muscle atrophy with dysfunctional proteins. d HD is characterized by including mutant Huntingtin protein (mHTT) and degeneration of medium spiny neurons in the basal ganglia, and corpus striatum of the brain. AD Alzheimer’s disease, ALS amyotrophic lateral sclerosis, BDNF brain-derived neurotrophic factor, EAL endosomal-autophagic-lysosomal pathway, GABA gamma-aminobutyric acid, HD Huntington’s disease, PSEN presenilin 1, SNCA synuclein alpha.


