[ad_1]

A team at the University of Missouri has devised a method to eliminate most nanoplastics from water using eco-friendly solvents, suitable for both fresh and saltwater applications.
Nanoplasticst are an emerging enemy of human health. Much smaller in size than the diameter of an average human hair, nanoplastics are invisible to the naked eye.
Linked to cardiovascular and respiratory diseases in people, nanoplastics continue to build up, largely unnoticed, in the world’s bodies of water. The challenge remains to develop a cost-effective solution to get rid of nanoplastics while leaving clean water behind.
Now, researchers at the University of Missouri have developed a revolutionary liquid-based solution that eliminates more than 98% of these microscopic plastic particles from water. This method, detailed in new study published in ACS Applied Engineering Materials, promises significant advancements in water purification technology.

“Nanoplastics can disrupt aquatic ecosystems and enter the food chain, posing risks to both wildlife and humans,” said Piyuni Ishtaweera, a recent alumna who led the study while earning her doctorate in nano and materials chemistry at Mizzou. “In layman’s terms, we’re developing better ways to remove contaminants such as nanoplastics from water.”
Innovative Purification Methods
The novel method — using water-repelling solvents made from natural ingredients — not only offers a practical solution to the pressing issue of nanoplastic pollution but also paves the way for further research and development in advanced water purification technologies.

“Our strategy uses a small amount of designer solvent to absorb plastic particles from a large volume of water,” said Gary Baker, an associate professor in Mizzou’s Department of Chemistry and the study’s corresponding author. “Currently, the capacity of these solvents is not well understood. In future work, we aim to determine the maximum capacity of the solvent. Additionally, we will explore methods to recycle the solvents, enabling their reuse multiple times if necessary.”
Scaling and Future Applications
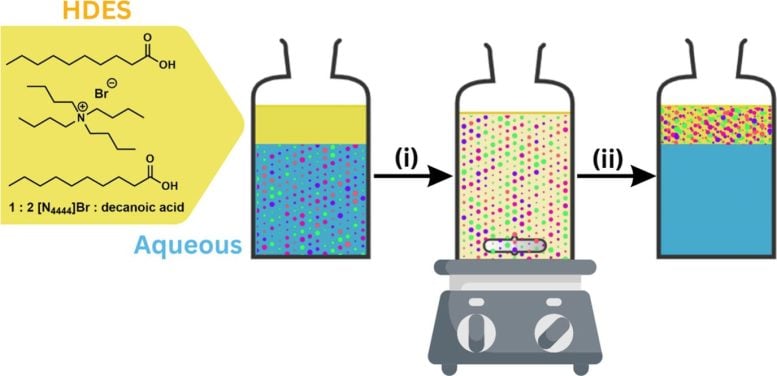
Initially, the solvent sits on the water’s surface the way oil floats on water. Once mixed with water and allowed to reseparate, the solvent floats back to the surface, carrying the nanoplastics within its molecular structure.
In the lab, the researchers simply use a pipette to remove the nanoplastic-laden solvent, leaving behind clean, plastic-free water. Baker said future studies will work to scale up the entire process so that it can be applied to larger bodies of water like lakes and, eventually, oceans.
Implications and Next Steps
Ishtaweera, who now works at the U.S. Food and Drug Administration in St. Louis, noted that the new method is effective in both fresh and saltwater.
“These solvents are made from safe, non-toxic components, and their ability to repel water prevents additional contamination of water sources, making them a highly sustainable solution,” she said. “From a scientific perspective, creating effective removal methods fosters innovation in filtration technologies, provides insights into nanomaterial behavior and supports the development of informed environmental policies.”
The Mizzou team tested five different sizes of polystyrene-based nanoplastics, a common type of plastic used in the making of Styrofoam cups. Their results outperformed previous studies that largely focused on just a single size of plastic particles.
Reference: “Nanoplastics Extraction from Water by Hydrophobic Deep Eutectic Solvents” by Piyuni Ishtaweera, Colleen L. Ray, Wyland Filley, Garrett Cobb and Gary A. Baker, 4 June 2024, ACS Applied Engineering Materials.
DOI: 10.1021/acsaenm.4c00159
[ad_2]
Source link






















