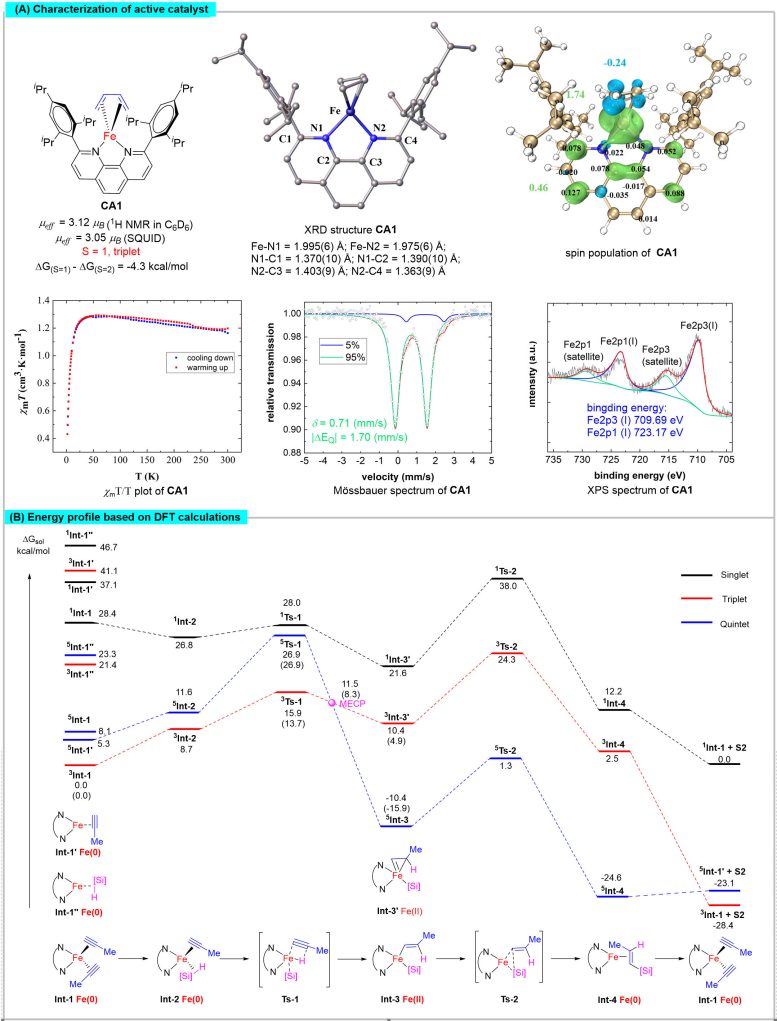
[ad_1]
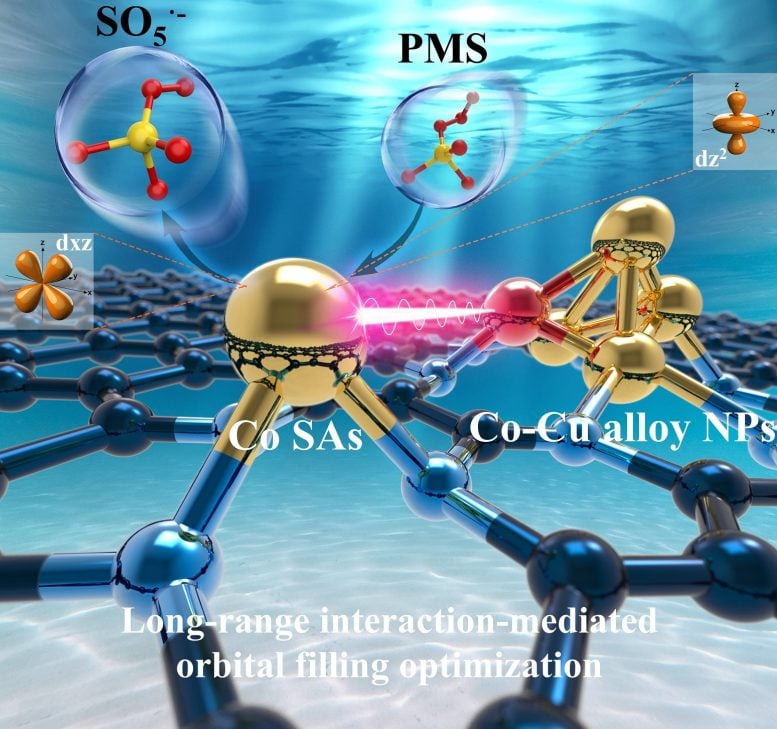
Cu heteroatoms in Co nanoparticles−mediated long−range interaction regulates dz2/dxz orbital occupancy of satellite Co single atom to enhance Fenton−like reaction. Credit: Science China Press
Scientists have developed Co-Cu alloy nanoparticles that efficiently activated chemical reactions for pollutant degradation, outperforming traditional methods and providing insights into atomic-level interactions in catalysts.
Recent research has focused on the combined effects of atomic clusters (ACs) or nanoparticles (NPs) and single atoms. However, prior studies have typically concentrated on basic interactions between pure NPs and single atoms (SAs), offering a limited perspective. They have not fully explored how modifications to NPs or ACs could enhance the overall activity of the complete metal NPs/ACs@M-N-C entity. Consequently, there is still much to learn about the precise construction and performance enhancement of the entire metal NPs/ACs@M-N-C structure.
In a new study, Co-Cu alloy NPs@Co-N-C (CC@CNC) was constructed using Co/Cu-modified zeolitic imidazolate framework-8 (ZIF-8) as precursor, followed by the pyrolysis and acid etching. The incorporation of Cu-induced formation of Co-Cu alloy NPs, originating from the low Tammann temperature of Cu, which was verified by the negative cohesive energy of Co10Cu3 (−0.06454, intending to aggregation) compared to that of Co13 (+1.690995, intending to dispersion).
Enhanced Efficiency in Chemical Reactions
The Co-Cu alloy NPs-supported Co SAs showed superior PMS activation efficiency compared to Co NPs-supported Co SAs (Co NPs@Co-N-C, C@CNC), evidenced by the decreased energy barriers of PMS adsorption (increased Co–O bond length)/PMS oxidation (increased O-H bond length and decreased O-O bond length) and SO5·- desorption (increased Co-O bond length), through optimizing dz2-O (PMS) and dxz-O (SO5·-) interaction.
Therefore, the CC@CNC highly efficiently removed 80.67% of 20 mg/L carbamazepine (CBZ) within 5 min, which was superior to the C@CNC counterpart (58.99% within 5 min). The serial quasi in situ techniques indicated the occurred PMS oxidation reaction on Co SAs, which can selectively generate 1O2 to effectively eliminate CBZ. This study can lay a solid foundation for the performance optimization strategy and underlying mechanism revelation in metal multiple-atom assembly@metal SAs catalysts at the atomic orbital level.
Reference: “Cu-optimized long-range interaction between Co nanoparticles and Co single atoms: Improved Fenton-like reaction activity” by Fan Mo, Zelin Hou, Qixing Zhou, Xixi Chen, Weitao Liu, Wendan Xue, Qi Wang, Jianling Wang, Tong Zheng and Zongxin Tao, 11 May 2024, Science Bulletin.
DOI: 10.1016/j.scib.2024.05.002
[ad_2]
Source link