[ad_1]
Antibiotics can reduce diversity in the gut microbiome, raising the risk of infections that cause diarrhoea – and the effects may last years
[ad_2]
Source link


[ad_1]
Antibiotics can reduce diversity in the gut microbiome, raising the risk of infections that cause diarrhoea – and the effects may last years
[ad_2]
Source link
[ad_1]
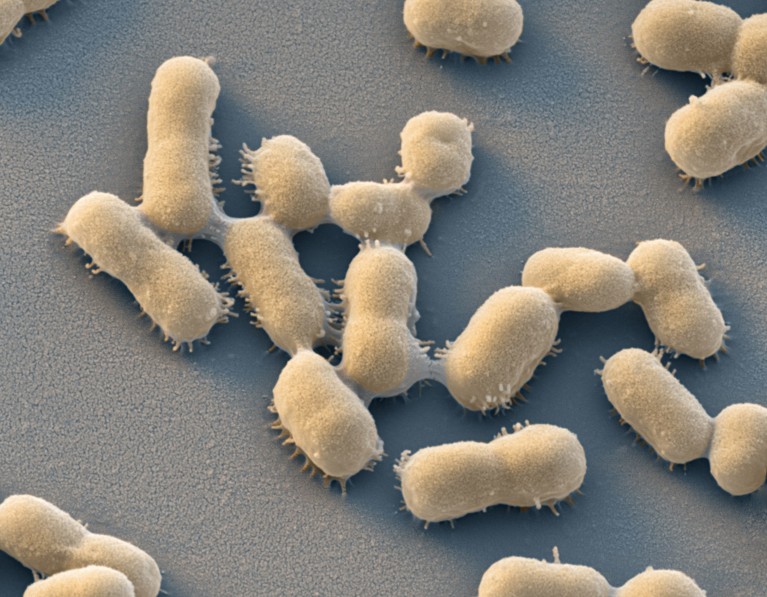
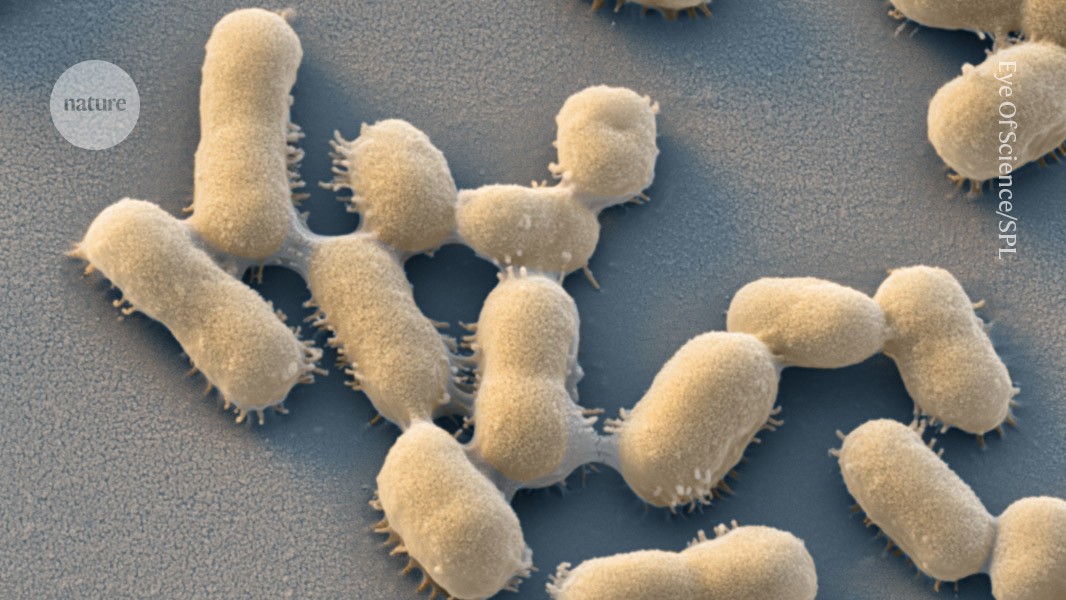
Acinetobacter baumannii — a bacterium associated with hospital-acquired infections that can develop resistance to antibiotics.Credit: Eye Of Science/Science Photo Library
More than 39 million people will die from antibiotic-resistant infections between now and 2050, according to an in-depth global analysis of antimicrobial resistance.
The report, published on 16 September in The Lancet1, found that between 1990 and 2021, more than one million people died from drug-resistant infections each year, and this could increase to nearly 2 million by 2050. Around 92 million lives could be saved between 2025 and 2050 with wider access to appropriate antibiotics and better treatment of infections, the report estimates.

The fight against antimicrobial resistance
“This is an important contribution for understanding how we’ve gotten where we are, and for giving a rational expectation of the future burden of [resistance] in order to inform next steps that can be undertaken,” says Joseph Lewnard, an epidemiologist at the University of California, Berkeley.
“I think the burden numbers are probably much higher than what has been reported here,” particularly in countries where there are data gaps, says Timothy Walsh, a microbiologist at the University of Oxford, UK. The figures suggest that the world is failing to meet the United Nation’s target of reducing mortality caused by antimicrobial resistance by 2030.
Researchers analysed mortality data and hospital records from 204 countries between 1990 and 2021, focusing on 22 pathogens, 84 combinations of bacteria and drugs they are resistant to and 11 diseases, including blood infections and meningitis.
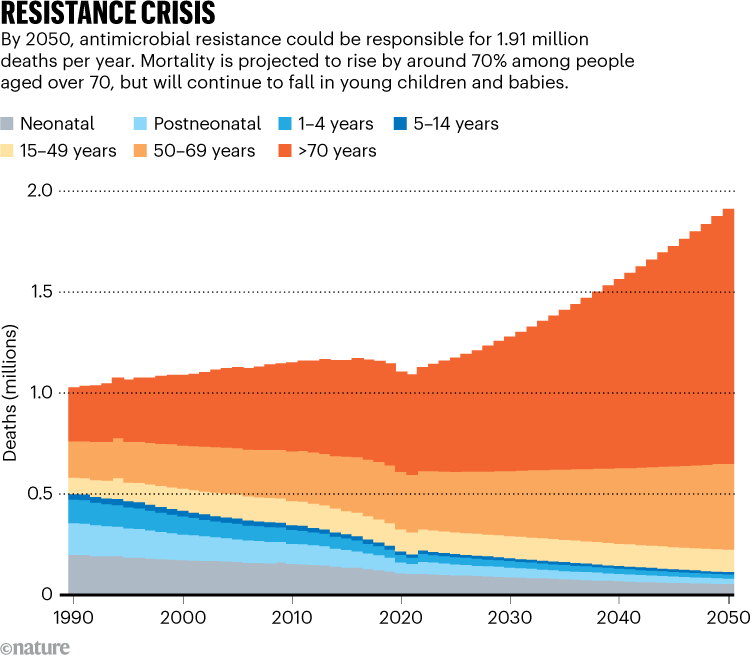
Their findings reveal that although the number of children aged under 5 dying from drug-resistant infections has fallen by more 50% over the past 3 decades, mortality rates in people aged over 70 have increased by 80% (see ‘Resistance crisis’).
Deaths from infections by Staphylococcus aureus — which infects skin, blood and internal organs — saw the biggest rise, increasing by 90.29%.
Source: Ref. 1
Many of the deadliest infections between 1990 and 2021 were caused by a group of bacteria with particularly strong drug resistance, called gram-negative bacteria. This category includes Escherichia coli and Acinetobacter baumannii — a pathogen associated with hospital-acquired infections.
Gram-negative bacteria are resistant to Carbapenem drugs, a class of antibiotics used to treat severe infections, and they can exchange antibiotic-resistance genes with different species as well as passing them to offspring. Deaths linked to Carbapenem-resistant gram-negative bacteria have risen by 149.51%, from 50,900 cases in 1990 to 127,000 cases in 2021.
The report estimates that, by 2050, antimicrobial resistance could cause 1.91 million deaths each year, and that a further 8.22 million people will die from illnesses associated with resistance. More than 65% of deaths attributed to AMR in 2050 will be among people aged over 70.
“This study shows we have a problem in health-system quality and prevention of infections,” says co-author Mohsen Naghavi, a physician and epidemiologist at the University of Washington in Seattle.
Regions with the highest predicted mortality rates include south Asia, Latin America and the Caribbean, and researchers emphasize that any strategies to tackle drug resistance must prioritize low- and middle-income countries.
Antibiotic resistance is a growing threat — is climate change making it worse?
“We need more global investment and a lot more real interactive engagement with low-income countries to make sure that they are equipped,” says Walsh. Strategies must ensure that hospitals in low-income countries have access to diagnostic tools, antibiotics, clean water and sanitation, he adds.
“Most of these deaths do not actually require new or boutique interventions to be prevented. That’s an important story that they tell,” says Lewnard.
Policymakers should also address the overuse of antibiotics in farming, which speeds up bacterial resistance, and invest in research for innovative antibiotic drugs, Walsh says.
The authors hope the report will “guide information on how to develop new drugs, which new drugs to focus on, what new vaccines to focus on”, says co-author Eve Wool, a research manager at the Institute for Health Metrics and Evaluation in Seattle, Washington.
[ad_2]
Source link

[ad_1]
This month, world leaders are gathering at the 79th session of the United Nations General Assembly in New York to discuss the problem of bacteria, fungi and protozoans increasingly evolving resistance to antimicrobial drugs. Historically, solutions have focused on high-income countries, even though 4.3 million of the estimated 5 million deaths associated with infections of drug-resistant bacteria occur in low- and middle-income countries. Ahead of the UN meeting, four specialists — from Bangladesh, Brazil, Nigeria and the Middle East — told Nature which changes will be most crucial where they live.

Senjuti Saha sees the need for a global commitment to support data collection on resistance.Credit: Vivan Mehra
For more than a decade, I have seen the impact of antimicrobial resistance (AMR) on people, particularly newborns, at first hand. Bangladesh Shishu Hospital and Institute in Dhaka — one of the collaborating organizations of the Child Health Research Foundation, where I work — is the country’s largest children’s hospital. Here, six out of every ten babies with infections caused by multidrug-resistant Klebsiella pneumoniae bacteria die, often just a few days after being infected.
In Bangladesh, a major difficulty in the fight against AMR is the lack of data on which bacteria are infecting whom, on how the various strains respond (or not) to which antibiotics and on how decisions are made about which drugs to use.

Stop delaying action on antimicrobial resistance — it is achievable and affordable
Bangladesh’s health-care system is unusual in that most people receive treatment in community clinics, diagnostic centres and pharmacies rather than in hospitals. People tend to go to hospitals only when treatment obtained from these other places fails. But because most of the country’s AMR data come from hospitals, government officials, clinicians and microbiologists have a skewed understanding of the problem.
Thanks to biases in the available data, many people think, for instance, that resistance is ubiquitous. Physicians often give patients late-generation or last-resort antibiotics even though early-generation drugs could still work. This in itself might be worsening resistance.
Another issue that affects Bangladesh in particular is the lack of protocols and basic interventions in and around health care that, if implemented, would greatly improve the situation.
For the babies at Shishu hospital, the most effective approach to saving lives is preventing infections from taking hold in the first place. Strategies that could substantially reduce AMR include investing in parental nutrition during gestation (which, in turn, would improve babies’ birth weights and reduce their susceptibility to infection), ensuring that pregnant people receive antenatal and perinatal care and promoting delivery practices that minimize the risk of babies (and birthing parents) getting infections.
What’s needed in Bangladesh — and in low- and middle-income countries more broadly — is a global commitment to support research and data collection on AMR, and investment in the basic interventions that will reduce it.

Ana Cristina Gales thinks that even small changes can be transformative for Brazil.Photo courtesy of Ana Cristina Gales
Five years ago, a 15-year-old boy was brought into the hospital where I work in São Paulo, Brazil, after developing an infection. He had cut his ankle while retrieving a kite from a stream. The boy developed a severe, systemic infection caused by methicillin-resistant Staphylococcus aureus and died around five weeks after being admitted.
That boy stuck in my mind, because he was around the age of my two daughters at the time. As a consultant in infectious diseases, I have seen too many people affected by AMR. One of the most frustrating aspects is that this devastating problem could be greatly reduced with relatively simple measures1.
Issues that affect hospitals in Brazil — and in many other countries — include staff members failing to keep surfaces and their hands clean, and failing to isolate people infected with bacteria that are resistant to antimicrobial drugs. Some people have to spend days waiting for beds on stretchers in hospital corridors, making it harder for health-care workers — who are often overworked — to implement basic measures to prevent and control infections. In fact, over the past two decades, investigators have described several cases in which identical antimicrobial-resistant bacteria (of the same clone) have been found in distinct hospitals2 — the implication being that resistant bacteria are being transferred between institutions, perhaps through the movement of patients.
But as illustrated by the case I described, AMR does not occur only in hospital settings. And in Brazil, one of the biggest challenges is people’s failure to recognize AMR as a public-health problem. Most people who don’t work in hospitals lack the knowledge needed to understand what AMR is and how it could affect their health. The country is battling endemic diseases, such as malaria, leishmaniasis and Chagas disease. And over the past 10 years, Brazil has experienced numerous epidemics caused by mosquito-borne viruses, including Zika, chikungunya, yellow fever, dengue and, most recently, Oropouche fever. Such diseases tend to receive more attention than does AMR, which is harder for people to pin to a particular cause.
Antibiotic resistance is a growing threat — is climate change making it worse?
As long as AMR is not seen as a significant health threat at a societal level, it will continue to be overlooked by policymakers and politicians. More positively, however, Brazil has enormous potential to reduce AMR, because it has a universal, decentralized health-care system in place for bringing medical interventions to everyone. It also has national programmes that provide people with vaccines and medications to treat HIV and hepatitis for free. Moreover, as one of the world’s largest producers of animal protein, Brazil could help to lead the reduction of antimicrobial use in livestock rearing. In fact, over the past decade or so, Brazil has already made some key advances when it comes to tackling AMR.
Since a law was introduced in 2010, people have been able to buy antibiotics (for human use) only with a medical prescription. Over the past few years, the government has invested millions of US dollars in research on AMR — and in surveillance, both in state laboratories and in hospitals. Furthermore, Brazil has developed a national plan to reduce the problem using a multisectoral and transdisciplinary ‘One Health’ approach, which is currently under review.
Transforming this plan into a successful programme, similar to the country’s vaccination, HIV and hepatitis schemes, will require more political buy-in and a lot more investment in efforts to raise public awareness. It will also require the integration of priorities across human, animal and environmental health. But clinicians, scientists, government officials and others don’t need access to advanced technologies and new antimicrobials to make an enormous difference. A few, relatively simple changes could be transformative: more parents and other caregivers vaccinating children; more people avoiding the use of antimicrobials when they are unnecessary, such as those used to treat viral respiratory infections; and expanding the nation’s basic sanitation programme, so that every Brazilian city treats its sewage before releasing it into the environment.

Improved access to health care can help to reduce antimicrobial resistance, says Iruka Okeke.Credit: Science for Africa Foundation
Most Nigerian people have a severe bacterial infection or malaria at least once a year. So they have a real as well as a perceived need for antimicrobials. But people are often unable to obtain the ones they need, or they use antimicrobials without advice from health-care providers. (In 2019, only 41% of Nigeria’s population could access health care easily.) Often, physicians don’t have access to the diagnostic tools needed to inform their decisions about what antimicrobials people should take, or individuals can’t afford those specific drugs. So, although in many settings the use of antimicrobials is excessive or inappropriate, in Nigeria, improving people’s access to health care and medications must be part of any plan to address AMR.
An article in the Lancet Series on Antimicrobial Resistance published in May1 proposes that authorities should aim to reduce the number of deaths caused by AMR by 10% and the inappropriate use of antibiotics in humans and other animals by 20% and 30%, respectively, by 2030. I am convinced that Nigeria could meet these targets using tools available today.
A patient receives dialysis in June at Al-Shifa hospital in Gaza City, which was damaged during the Israel–Hamas conflict.Credit: Dawoud Abu Alkas/Reuters
One-quarter of Nigeria’s population defecates outside, such as in fields, gutters and forests, instead of using a toilet, some or all of the time. And data from a 2024 report3 I was involved in show that most city residents rely on household water from wells or boreholes that is contaminated by faeces. This is the case in Ibadan, where I live. Agents of disease — including typhoid, diarrhoea and cholera — can spread easily, as can non-harmful (commensal) organisms harbouring resistance genes that can be transmitted to pathogenic bacteria. One such commensal microorganism is Escherichia coli, which is typically found in the intestine. Providing people with safe water and better sanitation could reduce the impact of both enteric infections (stomach or intestinal illnesses caused by microbes, such as viruses, bacteria and parasites) and AMR simultaneously, yielding an estimated return of US$5 or more for every $1 spent4.
Modest investment could also improve infection prevention and control practices at health-care facilities — such as promoting handwashing or the isolation of people infected with antimicrobial-resistant bacteria. In 2020, the Nigeria Centre for Disease Control and Prevention released updated guidelines on infection control and prevention. A modelling study5, published in May as part of the Lancet series, indicates that by following such guidelines judiciously, health-care providers could prevent antimicrobial-resistant infections in health-care settings, which currently account for about 300,000 deaths worldwide.

The fight against antimicrobial resistance
Nigeria has been rolling out pneumococcal vaccines since 2014 and rotavirus vaccines since 2022. But coverage, currently at 60% and 49%, respectively (according to data from the United Nations’ children’s charity UNICEF), needs to be extended. Malaria and typhoid-conjugate vaccines are yet to be deployed. Moreover, a global shortage of cholera vaccines has hampered their use in recent outbreaks. All of these vaccines could reduce levels of AMR — by lowering the chance of complications arising that involve bacterial infections, and of people administering antibiotics inappropriately. According to the modelling study in the Lancet series, vaccines could prevent around 180,000 deaths associated with AMR annually5.
In 2017, Nigeria launched an AMR surveillance system. Once this system covers a greater geographical area and more comprehensive data are collected — from people, animals and the environment — investigators will be able to quantify the impacts of each of these tools and support the prioritization of interventions for national deployment.

Humanitarian relief efforts should prioritize infection surveillance, says Nour Shamas.Credit: Aziz Reguig
I am constantly worrying about whether my mother, who has a recurrent infection of the urinary tract (which is resistant to most antibiotics), will be able to keep accessing the health-care services and antibiotics she needs. She lives in Beirut, Lebanon, and I am always reminding her to keep her passport and antibiotics to hand in case she has to flee in the face of a military attack.
The impacts of political and economic instability and conflict are often overlooked when people consider the factors contributing to AMR. But it’s much harder to implement and maintain the types of intervention often recommended to mitigate resistance, such as restrictions that limit over-the-counter dispensing of antimicrobials, in Lebanon and other eastern Mediterranean countries that are experiencing ongoing conflict. In fact, such an intervention could limit access to lifesaving antimicrobials for those who cannot otherwise obtain them.
The destruction of health-care facilities, schools and other infrastructure as well as people’s livelihoods, and the displacement of both local people and refugees, provide an ideal environment for AMR to increase. In those places facing conflict or receiving refugees, living spaces are becoming increasingly crowded. Many people have lost access to clean water and sanitation, health care, medications and vaccinations. Most have lost wages and many are no longer able to obtain adequate nutrition, and people are more likely to use antimicrobials inappropriately — if they can get hold of them.
Although investment must be made globally in interventions to mitigate resistance, I urge all funders and aid organizations to factor AMR into humanitarian relief efforts. More research must be done on the impact of AMR on communities living in conflict-affected areas, and on context-specific solutions. Extra resources must be devoted to improving supply chains and access to antimicrobials and diagnostic tools, and to improving their use in these regions. And AMR surveillance, infection control and health systems as a whole must be strengthened in conflict-affected areas.
[ad_2]
Source link

[ad_1]
The problem of antimicrobial resistance (AMR) — bacteria, fungi or protozoans evolving resistance to antimicrobial substances — is worsening. This month, heads of state are gathering in New York City at the 79th session of the United Nations General Assembly to discuss the issue, among other global challenges.
This is the second time that AMR has been featured at a high-level UN meeting. The first one, in 2016, highlighted the importance of the problem, which is associated with nearly five million deaths each year worldwide. Although there has been some progress in the past eight years, such as the development of national action plans by many countries, the pace of change has been slow. I am presenting at the upcoming meeting, and I hope to convince attendees that the next eight years could look very different.
Tackling antimicrobial resistance needs a tailored approach — four specialists weigh in
One of the main factors driving the global rise of AMR is the use of inappropriate or substandard antibiotics. Indeed, people in low- and middle-income countries, which often lack ‘second-line’ antibiotics — more effective, and often more expensive, than those that are the first choice for treatment — are much more likely to die of infections caused by resistant bacteria than are those in high-income nations. A modelling study published in The Lancet in May (which I was involved in) indicates, however, that even a fairly modest global investment — in the range of hundreds of millions of US dollars — to help prevent bacterial infections and improve access to relatively inexpensive antibiotics could avert millions of deaths (J. A. Lewnard et al. Lancet 403, 2439–2454; 2024).
Each year, about 7.7 million people die from bacterial infections. One-fifth of these are children under the age of five. According to our Lancet study, at least 750,000 of these deaths could be averted through prevention strategies such as providing safe water and good sanitation, ensuring that children receive recommended vaccines, isolating individuals infected with resistant bacteria and implementing protocols to increase the frequency of handwashing in hospitals. Our study also indicates that the largest reduction in deaths would come from improving people’s access to antibiotics that are not available in many countries where most of the world’s bacterial-infection-related deaths occur.
Antibiotic resistance is a growing threat — is climate change making it worse?
Malaria and most bacterial infections do not last as long as do tuberculosis or AIDS, from which people tend to die months or years after infection. A child with an infection who develops a fever in the morning can be dead the next day if they don’t receive the right antibiotics. But in low- and middle-income countries, those are unavailable in many public-sector clinics. Parents and other carers must frequently turn to their local pharmacies for help. Because it is harder in those pharmacies than in public-sector ones to set up systems that limit the entry of inappropriate, poor-quality or fake drugs, hundreds of millions of people are using them.
Bacterial infections have been unfairly neglected — probably because a diverse array of pathogens are responsible for them, so there is no clear group of stakeholders who can advocate for change, as is the case for AIDS, tuberculosis and malaria. Each year, the Global Fund to Fight AIDS, Tuberculosis and Malaria, based in Geneva, Switzerland, spends nearly US$5 billion to ensure that the drugs to treat these diseases are available in low-income countries. These three diseases collectively kill about 2.8 million people each year — less than half the number linked to bacterial infections other than tuberculosis.

The fight against antimicrobial resistance
International funders, such as the Global Fund, must step up. People with HIV have a high risk of developing bacterial, viral, fungal and protozoal infections. Providing people with access to effective diagnostics and antibiotics targeting bacterial infections more broadly would be a natural extension of the Global Fund’s existing mandate.
Furthermore, prevention strategies — especially the provision of vaccines, safe water and good sanitation — need to be supported by organizations such as Gavi, the Vaccine Alliance, as well as through bilateral donors, including the United States Agency for International Development in Washington DC. They also need to be prioritized in national budgets in low- and middle-income countries.
Experience from past UN declarations shows that specific commitments made by countries are more likely to translate into action than is language that is unclear about what is expected of which organizations. The Lancet Series on Antimicrobial Resistance, published in May, calls for a 10% reduction in human mortality from AMR, a 20% reduction in the inappropriate antibiotic use in people and a 30% reduction in the inappropriate antibiotic use in animals by 2030 (see go.nature.com/4d4xg) — all relative to levels in 2019. And support is growing for an independent panel set up by four intergovernmental organizations — the World Health Organization, the United Nations Environment Programme, the Food and Agriculture Organization and the World Organisation for Animal Health — to appraise the evidence around tackling AMR.
With investment from global funders, specific targets and accountability through an independent panel, there is a much higher chance of this year’s discussions at the General Assembly translating into global action to tackle AMR.
The author declares no competing interests.
[ad_2]
Source link
[ad_1]

Some microbes have a growing resistance to our antibiotics
iStockphoto
The number of global deaths directly attributable to antibiotic-resistant bacterial infections is forecast to rise from a record 1.27 million a year in 2019 to 1.91 million a year by 2050. In total, antibiotic resistance is expected to kill 39 million people between now and 2050 – but more than a third of that toll could be averted if we take action.
Resistance occurs when microbes evolve the ability to survive drugs that were deadly to them, meaning they no longer clear up infections. Because of the widespread use of antibiotics, in farming as well as healthcare, a growing number of microbes are becoming resistant and spreading globally, but the full scale of the problem is unclear.
To address this, Eve Wool at the Institute of Health Metrics and Evaluation (IHME) in Seattle and her colleagues have tried to estimate the annual number of deaths due to antibiotic resistance from 1990 to 2021. “Our estimates are based on more than 500 million records,” says Wool. “We have a lot of coverage geographically and across time.”
While the overall number of fatalities due to this has been rising, the team found that the figure for young children has been falling as a result of vaccinations and improved healthcare. Between 1990 and 2021, deaths due to antibiotic resistance decreased by more than 50 per cent among children younger than 5, compared with a rise of more than 80 per cent in adults over 70.
Overall, deaths attributable to antibiotic resistance rose from 1.06 million in 1990 to 1.27 million in 2019 and then fell to 1.14 million in 2021, the team concludes. However, the decline in 2020 and 2021 is thought to be a temporary blip caused by covid-19 control measures reducing other kinds of infections, too, rather than to a lasting improvement in combatting resistance.
In the study’s “most likely” scenario for the decades to come, deaths from antibiotic resistance rise to 1.91 million a year by 2050. In a scenario in which new antibiotics are developed against the most problematic bacteria, 11 million deaths would be averted between now and mid-century. In a “better care” scenario where more people also have access to good healthcare, even more deaths are avoided.
The 1.91 million annual deaths figure is much lower than an often cited one of 10 million deaths in 2050, from a 2016 review. That forecast was based on less reliable estimates and also included the problem of resistance to non-antibiotic drugs in diseases such as HIV and malaria, says team member Mohsen Naghavi, also at the IHME.
The new study is more thorough than previous efforts, says Marlieke de Kraker at Geneva University Hospitals in Switzerland, but still has some major limitations. For instance, it assumes the risk of antibiotic resistant infections causing deaths is the same around the world, when this isn’t the case. “If basic healthcare infrastructure is limited, drug-resistant infections do not necessarily lead to more deaths than drug-susceptible infections,” says de Kraker.
She is also sceptical about the team’s forecasts. “I feel predicting antimicrobial resistance trends is very unreliable,” says de Kraker. Drug-resistant versions of microbes can suddenly emerge or disappear without experts really understanding the underlying mechanisms, and there are frequently black swan events, which are impossible to predict, she says.
Topics:
[ad_2]
Source link

[ad_1]

Shewanella oneidensis bacteria could remove antibiotic resistance genes from wastewater
Ella Maru Studio/Science Photo Library
Bacteria engineered to destroy DNA can remove more than 99 per cent of genes that confer antibiotic resistance from wastewater. Treating wastewater with this method could help slow the spread of antibiotic resistance.
Disease-causing bacteria can absorb resistance genes that damaged or dead bacteria have released into the environment. This makes wastewater one of the largest environmental reservoirs for antibiotic resistance genes, says James Tiedje at Michigan State University. Affected microbes can spread to people through water, food or livestock.
[ad_2]
Source link
[ad_1]
Lewis, K. The science of antibiotic discovery. Cell 181, 29–45 (2020).
CAS
PubMed
Google Scholar
Tacconelli, E. et al. Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 18, 318–327 (2018).
Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 399, 629–655 (2022).
Ramos-Castaneda, J. A. et al. Mortality due to KPC carbapenemase-producing Klebsiella pneumoniae infections: systematic review and meta-analysis: mortality due to KPC Klebsiella pneumoniae infections. J. Infect. 76, 438–448 (2018).
Xu, L., Sun, X. & Ma, X. Systematic review and meta-analysis of mortality of patients infected with carbapenem-resistant Klebsiella pneumoniae. Ann. Clin. Microbiol. Antimicrob. 16, 18 (2017).
Zgurskaya, H. I., Rybenkov, V. V., Krishnamoorthy, G. & Leus, I. V. Trans-envelope multidrug efflux pumps of Gram-negative bacteria and their synergism with the outer membrane barrier. Res. Microbiol. 169, 351–356 (2018).
Staley, J. T. & Konopka, A. Measurement of in situ activities of nonphotosynthetic microorganisms in aquatic and terrestrial habitats. Annu. Rev. Microbiol. 39, 321–346 (1985).
CAS
PubMed
Google Scholar
D’Onofrio, A. et al. Siderophores from neighboring organisms promote the growth of uncultured bacteria. Chem. Biol. 17, 254–264 (2010).
Fenn, K. et al. Quinones are growth factors for the human gut microbiota. Microbiome 5, 161 (2017).
Strandwitz, P. et al. GABA-modulating bacteria of the human gut microbiota. Nat. Microbiol. 4, 396–403 (2019).
CAS
PubMed
Google Scholar
Ling, L. L. et al. A new antibiotic kills pathogens without detectable resistance. Nature 517, 455–459 (2015). This paper describes the discovery of teixobactin from an uncultured bacterium.
Shukla, R. et al. An antibiotic from an uncultured bacterium binds to an immutable target. Cell 186, 4059–4073.e4027 (2023).
CAS
PubMed
Google Scholar
Pantel, L. et al. Odilorhabdins, antibacterial agents that cause miscoding by binding at a new ribosomal site. Mol. Cell 70, 83–94 e87 (2018).
CAS
PubMed
Google Scholar
Imai, Y. et al. A new antibiotic selectively kills Gram-negative pathogens. Nature 576, 459–464 (2019).
Imai, Y. et al. Evybactin is a DNA gyrase inhibitor that selectively kills Mycobacterium tuberculosis. Nat. Chem. Biol. 18, 1236–1244 (2022). This paper describes the discovery of darobactins that target BamA in the outer membrane of Gram-negative bacteria.
Miller, R. D. et al. Computational identification of a systemic antibiotic for gram-negative bacteria. Nat. Microbiol. 7, 1661–1672 (2022).
Shahsavari, N. et al. A silent operon of Photorhabdus luminescens encodes a prodrug mimic of GTP. mBio 13, e0070022 (2022).
Libis, V. et al. Multiplexed mobilization and expression of biosynthetic gene clusters. Nat. Commun. 13, 5256 (2022). This paper describes an approach for efficient cloning of environmental DNA for the expression of BGCs.
Gavriilidou, A. et al. Compendium of specialized metabolite biosynthetic diversity encoded in bacterial genomes. Nat. Microbiol. 7, 726–735 (2022). This study catalogues BGCs from sequenced genomes and links them to taxonomy and biogeography.
CAS
PubMed
Google Scholar
O’Shea, R. & Moser, H. E. Physicochemical properties of antibacterial compounds: Implications for drug discovery. J. Med. Chem. 51, 2871–2878 (2008).
Richter, M. F. et al. Predictive compound accumulation rules yield a broad-spectrum antibiotic. Nature 545, 299–304 (2017). This study provides rules for compound penetration into Gram-negative bacteria.
Mehla, J. et al. Predictive rules of efflux inhibition and avoidance in Pseudomonas aeruginosa. mBio 12, e02785–20 (2021). This study analyses physico-chemical properties of compounds that enable penetration into P. aeruginosa, and synthesis of MDR inhibitors.
Zhao, S. et al. Defining new chemical space for drug penetration into Gram-negative bacteria. Nat. Chem. Biol. 16, 1293–1302 (2020).
Mansbach, R. A. et al. Machine learning algorithm identifies an antibiotic vocabulary for permeating Gram-negative bacteria. J. Chem. Inf. Model. 60, 2838–2847 (2020).
Geddes, E. J. et al. Porin-independent accumulation in Pseudomonas enables antibiotic discovery. Nature 624, 145–153 (2023).
Parker, E. N. et al. Implementation of permeation rules leads to a FabI inhibitor with activity against Gram-negative pathogens. Nat. Microbiol. 5, 67–75 (2020).
CAS
PubMed
Google Scholar
Durand-Reville, T. F. et al. Rational design of a new antibiotic class for drug-resistant infections. Nature 597, 698–702 (2021).
ADS
CAS
PubMed
Google Scholar
Srinivas, N. et al. Peptidomimetic antibiotics target outer-membrane biogenesis in Pseudomonas aeruginosa. Science 327, 1010–1013 (2010).
ADS
CAS
PubMed
Google Scholar
Luther, A. et al. Chimeric peptidomimetic antibiotics against Gram-negative bacteria. Nature 576, 452–458 (2019).
ADS
CAS
PubMed
Google Scholar
Pahil, K. S. et al. A new antibiotic traps lipopolysaccharide in its intermembrane transporter. Nature 625, 572–577 (2024).
Zampaloni, C. et al. A novel antibiotic class targeting the lipopolysaccharide transporter. Nature 625, 566–571 (2024).
Fleming, A. On the antibacterial action of cultures of a penicillium, with special reference to their use in the isolation of B. influenzae. Br. J. Exp. Pathol. 10, 226 (1929).
Mora-Ochomogo, M. & Lohans, C. T. β-Lactam antibiotic targets and resistance mechanisms: from covalent inhibitors to substrates. RSC Med. Chem. 12, 1623–1639 (2021).
Silver, L. L. Multi-targeting by monotherapeutic antibacterials. Nat. Rev. Drug Discov. 6, 41–55 (2007).
CAS
PubMed
Google Scholar
Reading, C. & Cole, M. Clavulanic acid: a beta-lactamase-inhibiting beta-lactam from Streptomyces clavuligerus. Antimicrob. Agents Chemother. 11, 852–857 (1977).
Levasseur, P. et al. Efficacy of a ceftazidime–avibactam combination in a murine model of septicemia caused by Enterobacteriaceae species producing ampc or extended-spectrum β-lactamases. Antimicrob. Agents Chemother. 58, 6490–6495 (2014).
Wunderink, R. G. et al. Effect and safety of meropenem-vaborbactam versus best-available therapy in patients with carbapenem-resistant Enterobacteriaceae infections: The TANGO II randomized clinical trial. Infect. Dis. Ther. 7, 439–455 (2018).
Lewis, K. & Ausubel, F. M. Prospects for plant-derived antibacterials. Nat. Biotechnol. 24, 1504–1507 (2006).
CAS
PubMed
Google Scholar
Stermitz, F. R., Lorenz, P., Tawara, J. N., Zenewicz, L. A. & Lewis, K. Synergy in a medicinal plant: antimicrobial action of berberine potentiated by 5′-methoxyhydnocarpin, a multidrug pump inhibitor. Proc. Natl Acad. Sci. USA 97, 1433–1437 (2000).
Moniruzzaman, M. et al. Analysis of orthogonal efflux and permeation properties of compounds leads to the discovery of new efflux pump inhibitors. ACS Infect. Dis. 8, 2149–2160 (2022).
Dominguez-Bello, M. G., Godoy-Vitorino, F., Knight, R. & Blaser, M. J. Role of the microbiome in human development. Gut 68, 1108–1114 (2019).
CAS
PubMed
Google Scholar
Schnizlein, M. K. & Young, V. B. Capturing the environment of the Clostridioides difficile infection cycle. Nat. Rev. Gastroenterol. Hepatol. 19, 508–520 (2022).
Anthony, W. E. et al. Acute and persistent effects of commonly used antibiotics on the gut microbiome and resistome in healthy adults. Cell Rep. 39, 110649 (2022).
Feuerstadt, P. et al. SER-109, an oral microbiome therapy for recurrent Clostridioides difficile infection. N. Engl. J. Med. 386, 220–229 (2022). This paper describes the introduction of a new type of ‘drug’—an assemblage of clostridial spores for the treatment of C. difficile infection.
CAS
PubMed
Google Scholar
Lopetuso, L. R., Scaldaferri, F., Petito, V. & Gasbarrini, A. Commensal clostridia: leading players in the maintenance of gut homeostasis. Gut Pathog. 5, 23 (2013).
Mikusova, K., Slayden, R. A., Besra, G. S. & Brennan, P. J. Biogenesis of the mycobacterial cell wall and the site of action of ethambutol. Antimicrob. Agents Chemother. 39, 2484–2489 (1995).
Chahine, E. B., Karaoui, L. R. & Mansour, H. Bedaquiline: a novel diarylquinoline for multidrug-resistant tuberculosis. Ann. Pharmacother. 48, 107–115 (2014).
Diallo, D. et al. Antituberculosis therapy and gut microbiota: review of potential host microbiota directed-therapies. Front. Cell. Infect. Microbiol. 11, 673100 (2021).
Quigley, J. et al. Novel antimicrobials from uncultured bacteria acting against Mycobacterium tuberculosis. mBio 11, e01516–e01520 (2020).
Motiwala, T., Mthethwa, Q., Achilonu, I. & Khoza, T. ESKAPE pathogens: looking at Clp ATPases as potential drug targets. Antibiotics 11, 1218 (2022).
Rempel, S. et al. A mycobacterial ABC transporter mediates the uptake of hydrophilic compounds. Nature 580, 409–412 (2020).
ADS
CAS
PubMed
Google Scholar
Leimer, N. et al. A selective antibiotic for Lyme disease. Cell 184, 5405–5418.e5416 (2021). This paper describes the identification of an antibiotic for selective action against B. burgdorferi.
Polikanov, Y. S., Melnikov, S. V., Soll, D. & Steitz, T. A. Structural insights into the role of rRNA modifications in protein synthesis and ribosome assembly. Nat. Struct. Mol. Biol. 22, 342–344 (2015).
Chatterjee, A. N. & Perkins, H. R. Compounds formed between nucleotides related to the biosynthesis of bacterial cell wall and vancomycin. Biochem. Biophys. Res. Commun. 24, 489–494 (1966).
CAS
PubMed
Google Scholar
Munch, D. & Sahl, H. G. Structural variations of the cell wall precursor lipid II in Gram-positive bacteria – Impact on binding and efficacy of antimicrobial peptides. Biochim. Biophys. Acta 1848, 3062–3071 (2015).
Leclercq, R., Derlot, E., Duval, J. & Courvalin, P. Plasmid-mediated resistance to vancomycin and teicoplanin in Enterococcus faecium. N. Engl. J. Med. 319, 157–161 (1988).
CAS
PubMed
Google Scholar
Marshall, C. G., Broadhead, G., Leskiw, B. K. & Wright, G. D. d-Ala–d-Ala ligases from glycopeptide antibiotic-producing organisms are highly homologous to the enterococcal vancomycin-resistance ligases VanA and VanB. Proc. Natl Acad. Sci. USA 94, 6480–6483 (1997).
Shukla, R. et al. Teixobactin kills bacteria by a two-pronged attack on the cell envelope. Nature 608, 390–396 (2022).
Shukla, R. et al. An antibiotic from an uncultured bacterium binds to an immutable target. Cell 186, 4059–4073 (2023).
CAS
PubMed
Google Scholar
Homma, T. et al. Dual targeting of cell wall precursors by teixobactin leads to cell lysis. Antimicrob. Agents Chemother. 60, 6510–6517 (2016).
Schatz, A., Bugie, E. & Waksman, S. A. Streptomycin, a substance exhibiting antibiotic activity against Gram-positive and Gram-negative bacteria. Proc. Soc. Exp. Biol. Med. 55, 66–69 (1944).
CAS
Google Scholar
Wilson, D. N. Ribosome-targeting antibiotics and mechanisms of bacterial resistance. Nat. Rev. Microbiol. 12, 35–48 (2014).
CAS
PubMed
Google Scholar
Aguirre Rivera, J. et al. Real-time measurements of aminoglycoside effects on protein synthesis in live cells. Proc. Natl Acad. Sci. USA 118, e2013315118 (2021).
Andersson, D. I., Bohman, K., Isaksson, L. A. & Kurland, C. G. Translation rates and misreading characteristics of rpsD mutants in Escherichia coli. Mol. Genetics Genomics 187, 467–472 (1982).
CAS
Google Scholar
Wohlgemuth, I. et al. Translation error clusters induced by aminoglycoside antibiotics. Nat. Commun. 12, 1830 (2021). This study reveals the basis of killing by aminoglycosides—the introduction of strings of errors into nascent proteins.
Tokuriki, N. & Tawfik, D. S. Stability effects of mutations and protein evolvability. Curr. Opin. Struct. Biol. 19, 596–604 (2009).
CAS
PubMed
Google Scholar
Kling, A. et al. Targeting DnaN for tuberculosis therapy using novel griselimycins. Science 348, 1106–1112 (2015).
ADS
CAS
PubMed
Google Scholar
Lewis, K. (ed.) Persister Cells and Infectious Disease (Springer Nature, 2019).
Dorr, T., Vulic, M. & Lewis, K. Ciprofloxacin causes persister formation by inducing the TisB toxin in Escherichia coli. PLoS Biol. 8, e1000317 (2010).
Berghoff, B. A., Hoekzema, M., Aulbach, L. & Wagner, E. G. Two regulatory RNA elements affect TisB-dependent depolarization and persister formation. Mol. Microbiol. 103, 1020–1033 (2017).
CAS
PubMed
Google Scholar
Romilly, C., Deindl, S. & Wagner, E. G. H. The ribosomal protein S1-dependent standby site in tisB mRNA consists of a single-stranded region and a 5′ structure element. Proc. Natl Acad. Sci. USA 116, 15901–15906 (2019).
Schumacher, M. A. et al. HipBA–promoter structures reveal the basis of heritable multidrug tolerance. Nature 524, 59–64 (2015).
Manuse, S. et al. Bacterial persisters are a stochastically formed subpopulation of low-energy cells. PLoS Biol. 19, e3001194 (2021).
Quigley, J. & Lewis, K. Noise in a metabolic pathway leads to persister formation in Mycobacterium tuberculosis. Microbiol. Spectr. 10, e0294822 (2022).
Fleck, L. E. et al. A screen for and validation of prodrug antimicrobials. Antimicrob. Agents Chemother. 58, 1410–1419 (2014).
Goodreid, J. D. et al. Total synthesis and antibacterial testing of the A54556 cyclic acyldepsipeptides isolated from Streptomyces hawaiiensis. J. Nat. Prod. 77, 2170–2181 (2014).
CAS
PubMed
Google Scholar
Thomy, D. et al. The ADEP biosynthetic gene cluster in Streptomyces hawaiiensis NRRL 15010 reveals an accessory clpP gene as a novel antibiotic resistance factor. Appl. Environ. Microbiol. 85, e01292–19 (2019).
Brotz-Oesterhelt, H. et al. Dysregulation of bacterial proteolytic machinery by a new class of antibiotics. Nat. Med. 11, 1082–1087 (2005). This paper describes the discovery of the mechanism of killing by ADEP: dysregulation of the bacterial protease ClpP.
Olivares, A. O., Nager, A. R., Iosefson, O., Sauer, R. T. & Baker, T. A. Mechanochemical basis of protein degradation by a double-ring AAA+ machine. Nat. Struct. Mol. Biol. 21, 871–875 (2014).
Vahidi, S. et al. Reversible inhibition of the ClpP protease via an N-terminal conformational switch. Proc. Natl Acad. Sci. USA 115, E6447–E6456 (2018).
Griffith, E. C. et al. Ureadepsipeptides as ClpP Activators. ACS Infect. Dis. 5, 1915–1925 (2019).
Malik, I. T. et al. Functional characterisation of ClpP mutations conferring resistance to acyldepsipeptide antibiotics in firmicutes. ChemBioChem 21, 1997–2012 (2020).
Gatsogiannis, C., Balogh, D., Merino, F., Sieber, S. A. & Raunser, S. Cryo-EM structure of the ClpXP protein degradation machinery. Nat. Struct. Mol. Biol. 26, 946–954 (2019).
Ripstein, Z. A., Vahidi, S., Houry, W. A., Rubinstein, J. L. & Kay, L. E. A processive rotary mechanism couples substrate unfolding and proteolysis in the ClpXP degradation machinery. eLife 9, e52158 (2020).
Fei, X. et al. Structures of the ATP-fueled ClpXP proteolytic machine bound to protein substrate. eLife 9, e52774 (2020).
Sass, P. et al. Antibiotic acyldepsipeptides activate ClpP peptidase to degrade the cell division protein FtsZ. Proc. Natl Acad. Sci. USA 108, 17474–17479 (2011).
Silber, N., Mayer, C., Matos de Opitz, C. L. & Sass, P. Progression of the late-stage divisome is unaffected by the depletion of the cytoplasmic FtsZ pool. Commun. Biol. 4, 270 (2021).
Conlon, B. P. et al. Activated ClpP kills persisters and eradicates a chronic biofilm infection. Nature 503, 365–370 (2013). This paper describes the anti-persister activity of ADEP.
Mroue, N. et al. Pharmacodynamics of ClpP-activating antibiotic combinations against Gram-positive pathogens. Antimicrob. Agents Chemother. 64, e01554-19 (2019).
Brown Gandt, A. et al. In vivo and in vitro effects of a ClpP-activating antibiotic against vancomycin-resistant enterococci. Antimicrob. Agents Chemother. 62, e00424-18 (2018).
Brotz-Oesterhelt, H. & Vorbach, A. Reprogramming of the caseinolytic protease by ADEP antibiotics: molecular mechanism, cellular consequences, therapeutic potential. Front. Mol. Biosci. 8, 690902 (2021).
Frees, D., Gerth, U. & Ingmer, H. Clp chaperones and proteases are central in stress survival, virulence and antibiotic resistance of Staphylococcus aureus. Int. J. Med. Microbiol. 304, 142–149 (2014).
CAS
PubMed
Google Scholar
Illigmann, A., Thoma, Y., Pan, S., Reinhardt, L. & Brotz-Oesterhelt, H. Contribution of the Clp protease to bacterial survival and mitochondrial homoeostasis. Microb. Physiol. 31, 260–279 (2021).
CAS
PubMed
Google Scholar
Schuster, M. et al. Peptidomimetic antibiotics disrupt the lipopolysaccharide transport bridge of drug-resistant Enterobacteriaceae. Sci. Adv. 9, eadg3683 (2023).
Nguyen, H. et al. Characterization of a radical SAM oxygenase for the ether crosslinking in darobactin biosynthesis. J. Am. Chem. Soc. 144, 18876–18886 (2022).
Kaur, H. et al. The antibiotic darobactin mimics a β-strand to inhibit outer membrane insertase. Nature 593, 125–129 (2021).
ADS
CAS
PubMed
Google Scholar
Groß, S. et al. Improved broad-spectrum antibiotics against Gram-negative pathogens via darobactin biosynthetic pathway engineering. Chem. Sci. 12, 11882–11893 (2021).
Seyfert, C. E. et al. Darobactins exhibiting superior antibiotic activity by Cryo-EM structure guided biosynthetic engineering. Angew. Chem. Int. Ed. Engl. 62, e202214094 (2022).
Lin, Y. C. et al. Atroposelective total synthesis of darobactin A. J. Am. Chem. Soc. 144, 14458–14462 (2022).
Nesic, M. et al. Total synthesis of darobactin A. J. Am. Chem. Soc. 144, 14026–14030 (2022).
CAS
PubMed
Google Scholar
Tan, Y. S., Lane, D. P. & Verma, C. S. Stapled peptide design: principles and roles of computation. Drug Discov. Today 21, 1642–1653 (2016).
CAS
PubMed
Google Scholar
Maeda, K., Osato, T. & Umezawa, H. A new antibiotic, azomycin. J. Antibiot. 6, 182 (1953).
CAS
Google Scholar
Nakamura, S. Structure of azomycin, a new antibiotic. Pharm. Bull. 3, 379–383 (1955).
CAS
PubMed
Google Scholar
Shoji, J. H. et al. Isolation of azomycin from Pseudomonas fluorescens. J. Antibiot. 42, 1513–1514 (1989).
CAS
Google Scholar
Gupta, R. et al. Functionalized nitroimidazole scaffold construction and their pharmaceutical applications: a 1950–2021 comprehensive overview. Pharmaceuticals 15, 561 (2022).
Goldstein, B. P. et al. The mechanism of action of nitro-heterocyclic antimicrobial drugs. Metabolic activation by micro-organisms. J. Gen. Microbiol. 100, 283–298 (1977).
CAS
PubMed
Google Scholar
Miller, M. J. & Liu, R. Design and syntheses of new antibiotics inspired by nature’s quest for iron in an oxidative climate. Acc. Chem. Res. 54, 1646–1661 (2021).
Sato, T. & Yamawaki, K. Cefiderocol: discovery, chemistry, and in vivo profiles of a novel siderophore cephalosporin. Clin. Infect. Dis. 69, S538–S543 (2019). This paper describes the creation of an approved chimeric antibiotic utilizing a siderophore moiety for penetration into the cell.
Broder, S. The development of antiretroviral therapy and its impact on the HIV-1/AIDS pandemic. Antiviral Res. 85, 1–18 (2010).
CAS
PubMed
Google Scholar
Sanger, F., Nicklen, S. & Coulson, A. R. DNA sequencing with chain-terminating inhibitors. Proc. Natl Acad. Sci. USA 74, 5463–5467 (1977).
Wu, S. et al. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nat. Med. 15, 1016–1022 (2009).
Cao, Y. et al. Commensal microbiota from patients with inflammatory bowel disease produce genotoxic metabolites. Science 378, eabm3233 (2022).
Lee, J. Y., Tsolis, R. M. & Baumler, A. J. The microbiome and gut homeostasis. Science 377, eabp9960 (2022).
CAS
PubMed
Google Scholar
Cook, M. A. & Wright, G. D. The past, present, and future of antibiotics. Sci. Transl. Med. 14, eabo7793 (2022).
CAS
PubMed
Google Scholar
Smith, P. A. et al. Optimized arylomycins are a new class of Gram-negative antibiotics. Nature 561, 189–194 (2018).
ADS
CAS
PubMed
Google Scholar
[ad_2]
Source link
[ad_1]

Shutterstock/Kateryna Kon
When Jeanne Marrazzo was announced as director of the US National Institute of Allergy and Infectious Diseases (NIAID) last year, she became one of the leading public health decision-makers in the world. Replacing Anthony Fauci, whose tenure during the covid-19 pandemic made him a household name, the stakes have never been higher for the agency’s newest boss.
Having spent decades working across HIV prevention and sexually transmitted diseases, as well as overseeing therapeutic interventions for covid-19, Marrazzo is now in charge of NIAID’s $6.6 billion annual budget – and the future of the US response to infectious disease.
That involves working with the institute’s 21 laboratories across the country, leading the fight against ebola and HIV and spearheading efforts to develop new vaccines, therapies, diagnostics and technologies.
At the top of Marrazzo’s to-do list is tackling antimicrobial resistance (AMR), or drug-resistant superbugs, which are predicted to cause 10 million deaths per year by 2050 at an annual cost of $1 trillion to the global economy. In May, the UK’s former chief medical officer warned that the rise of these pathogens could make the pandemic look “minor” and that the issue is more acute than climate change.
Our warming world is partly responsible for increasing rates of AMR, with shifting climate conditions around the globe helping bacteria such as Salmonella and cholera-causing Vibrio to survive – and evade our current antibiotic weaponry completely. Here, Marrazzo outlines the things we should perhaps fear most, as well as some promising developments on the horizon.
Charlotte Lytton: In what ways is the…
[ad_2]
Source link
[ad_1]
Department of Chemistry, University of York, Heslington, York, UK
Katie A. Read, Lucy J. Carpenter & Sarah J. Moller
School of Chemistry, University of Leeds, Leeds, UK
Anoop S. Mahajan, Dwayne E. Heard, Hilke Oetjen, Michael J. Pilling & John M. C. Plane
School of Earth and the Environment (SEE), University of Leeds, Leeds, UK
Mathew J. Evans & James B. McQuaid
Instituto Nacional de Meteorologia Geofísica (INMG), Delegação de São Vicente, Monte, Mindelo, Cape Verde
Bruno V. E. Faria & Luis Mendes
National Centre for Atmospheric Science (NCAS), University of York, Heslington, York, UK
James R. Hopkins, James D. Lee & Alastair C. Lewis
Earth and Space Science Division, Jet Propulsion Laboratory, California Institute of Technology, Pasadena, California, USA
Alfonso Saiz-Lopez
[ad_2]
Source link

[ad_1]

Martin Rees at the 2017 Hay Festival of Literature in Hay on Wye, UK
Alamy Stock Photo
The Oscars. The Booker prize. The Nobels. The award ceremonies that punctuate our year are all inherently backward-looking, celebrating past achievements. But there is another type of award, one that looks to the future – the challenge prize. Such prizes don’t recognise past successes, rather incentivise future ones.
The idea is simple: a challenge is selected – with a clear-cut target – and a jackpot is offered to whoever first reaches that goal. Examples include the Longitude Prize on Antimicrobial Resistance (AMR), which has…
[ad_2]
Source link