The Jewish General Hospital and the Integrated Health and Social Services University Network for West-Central Montreal are committed to enhancing patient care through healthcare innovation. Here, they discuss their recent efforts to guide the institution towards a user-centric and technology-driven future.
TO FULLY appreciate the deep, long-term commitment to healthcare innovation at the Jewish General Hospital (JGH), one must start by looking back to 1934, the year the hospital was launched. Within days of opening their doors, surgeons were already using a mechanised, adjustable operating table – the first of its kind in Canada – that was particularly effective in fracture cases since it allowed X-ray imaging to be performed on the spot.
Pursuit of innovation in healthcare
Since those early days, a great deal has changed. Back then, the JGH was an autonomous healthcare institution. In contrast, today, it is the central pillar in a network of 34 interconnected and interdependent facilities, known as the Integrated Health and Social Services University Network for West-Central Montreal (or, more commonly, CIUSSS West-Central Montreal).
What has not changed, however, is the hospital’s – and, by extension, the entire network’s – intense dedication to healthcare innovation and research. This dedication is driven by a determination to deliver exceptional care that focuses on and responds to the needs of individual patients, clients, and other healthcare users.
Far from being a recent product of the digital age, the pursuit of innovation is deeply embedded in the DNA of the Jewish General Hospital and CIUSSS West-Central Montreal, with roots that date all the way back to that early operating room.
Dr Lawrence Rosenberg, President and CEO of CIUSSS West-Central Montreal, said: “This drive for innovation has manifested itself not just in a willingness to embrace change, but in a desire to actively nurture creative thinking and evidence-based research.

“At the same time, we understand what should not be changed: Compassion for our patients, one-to-one contact with them, respect for their feelings, and a commitment to listening to their concerns. Though we take pride in improvements to our practices and technology, our obligation is to keep asking ourselves, ‘What more can we do?’”
The result, most recently, has been a spate of impressive and, on occasion, award-winning initiatives. These include C4, a Command Centre where dozens of screens provide staff with real-time, network-wide data about numerous aspects of patient care; the Hospital@Home programme, in which certain qualified patients return home, where a hospital-based care team continuously and remotely monitors them; and the Connected Health Record (now in its preliminary phase), which will enable staff to use a single application to quickly and easily review a full range of up-to-date medical information about any patient seen at any CIUSSS site.
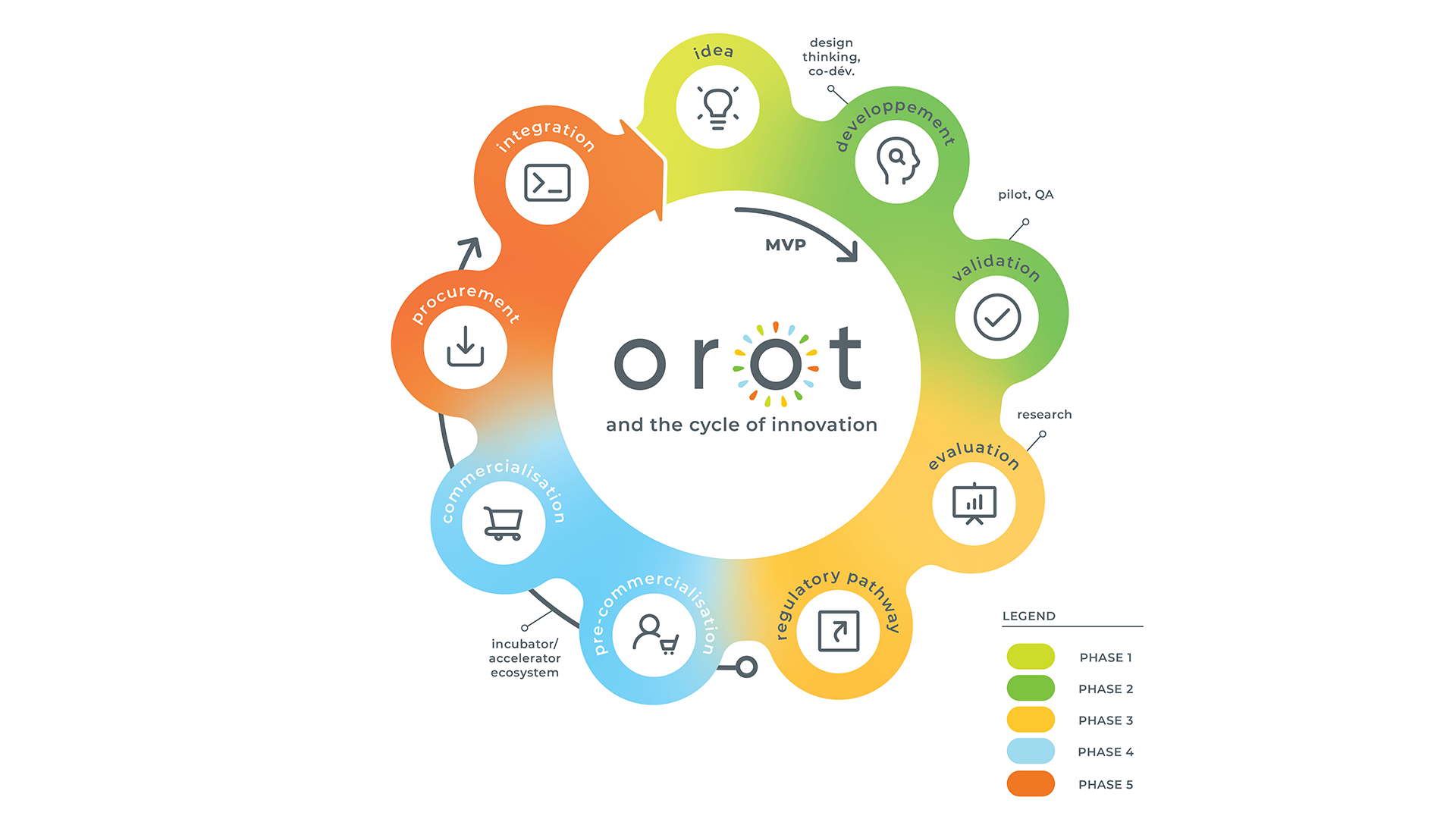
OROT
Of key importance in guiding the JGH and the CIUSSS in new directions is OROT (Hebrew for ‘illumination’), a connected health innovation hub and incubator.
With one of the most comprehensive and distinctive innovation programmes in a healthcare setting in Canada, OROT has a mandate to support organisation-wide efforts to reimagine the future of care through adaptive culture change, strategic foresight, digital transformation and the co-creation of technology.

Over the past four years, OROT has helped the CIUSSS leap toward a future where user-centric technology is assisting clinicians to provide more and better care, with improved results, to patients who are empowered to take charge of their health.
OROT’s many services are designed to support the network’s employees in reimagining the way they provide care. They do so by helping them gain knowledge and skills related to digital technology, leading them through the process of open, user-centric innovation, and offering them expertise in managing innovation and integrating technology.
To assist in building a future-proof healthcare system, OROT employs methods that involve strategic foresight to help the organisation consider how the trends that shape healthcare will affect the way that services must adapt in order to be resilient and thrive amid change.
In 2023, OROT facilitated two major cross-functional workshops to lead the directorates of CIUSSS West-Central Montreal through the process of future backcasting by equipping them with the means of anticipating and growing excited about taking the lead in transforming healthcare. Their goal was to re-imagine a primary-care centre and medical library of the future.
Through its internationally recognised incubation programme, OROT also fosters the creation and commercialisation of the next generation of digital health technologies. OROT offers a unique and integrated environment based on interdisciplinary collaboration and the process of co-creation and validation of new technologies. The aim is to catalyse their development, commercialisation, market launch, and, ultimately, user adoption.
To do this, OROT facilitates access and collaboration among health professionals, users, and entrepreneurs. Together, they cooperate in developing and implementing tools and products that satisfy the practical, real-world needs of patients and the requirements of the professionals who provide care.
The creation of innovative digital health technologies begins with an idea, which is tested, developed, and then commercialised. The process ends when the innovation – often in the form of a new tool or product – arrives in the hands of users, who may be clinicians, patients or caregivers. However, the journey to the marketplace can be very treacherous, as more than 95% of companies fail in the course of trying to serve the healthcare sector.
Danina Kapetanovic, the CIUSSS’s Chief Innovation Officer, Founder, and Head of OROT, notes that few companies succeed in developing and integrating technologies that truly transform healthcare because they use strategies that originate in the tech sector. As a result, companies start by inventing a product and then hope prospective consumers will find a good reason to buy it.
Ms Kapetanovic explained: “A better approach – our approach – is to support needs-driven innovation. As an initial step, we help entrepreneurs acquire a deep understanding of a particular problem in the healthcare sector. They gain this understanding through direct contact – facilitated by OROT – with healthcare professionals, patients and their families. This gives them a sound basis for designing technology that is uniquely suited to solving a problem.”
Working with entrepreneurs and experts from CIUSSS West-Central Montreal, OROT uses an integrated model of co-development, testing and evaluation, and implementation that is user-centric and informed by large-scale data science. The goal is to ensure that the proposed product will meet everyone’s requirements by the time it reaches the marketplace.
Utilising the OROT platform
Using this dynamic approach, OROT has made many unique endeavours possible. Among them (to name only a few):
Improving communication
Braver, a Québec startup that had developed an app for secure clinical communication, worked with OROT to enhance the platform to improve contact between clinicians and patients. Since early 2023, the app has been used in a paediatric setting in CIUSSS West-Central Montreal to give parents greater insight into the nature of their child’s therapy and to improve communication with therapists.
Harnessing AI
Partnering with IVADO Labs for their AI expertise and the CIUSSS’s Logistics Directorate, OROT has helped co-design and is currently testing an AI tool that will make logistics at the CIUSSS more accurate, cost-effective, efficient and proactive, thereby saving valuable resources.
Improving telephone services
OROT is collaborating with a CIUSSS directorate for proximity services and with a Québec company, VitrAi, by bringing in AI support to optimise the regional health consultation telephone service.
Optimised patient triage
In collaboration with the Toronto company Signal 1 and with the CIUSSS’s Hospital@Home programme, OROT is supporting the co-design of an algorithm that will allow for optimised patient triage.
Cutting-edge technologies
OROT has helped bring several cutting-edge technologies into the network. These include remote vital-sign monitoring devices currently used in the Hospital@Home programme, augmented reality to improve the care and stimulation of elderly individuals with Alzheimer’s disease, AI support to optimise patient scheduling in oncology clinics, sensors to optimise telerehabilitation services, and point-of-care blood and urine testing.
Comprehensive and tailored approach
Ms Kapetanovic points out that OROT is internationally recognised as a rarity among incubators. It guides and steers companies throughout the cycle of innovation while providing them with ongoing support and coaching until their products are integrated into the healthcare continuum.
This invaluable assistance – and its subsequent contribution to improving the quality of care – is the reason OROT has become an essential component of ‘Care Everywhere’, the broad-based, patient-centred approach to care that is fundamental to CIUSSS West-Central Montreal. As Dr Rosenberg notes, “Using ‘Care Everywhere’ as our guidepost, we continually strive to achieve the right outcomes by delivering the right care at the right time in the location that is most appropriate, safest and most convenient for patients and other healthcare users.” See OROT and the cycle of innovation graph pictured below.
Dedicated research facilities
Like innovation, research also has deep roots in the many facilities of CIUSSS West-Central Montreal, particularly at the Jewish General Hospital. During the JGH’s earliest decades, research was conducted in any adequate corner that happened to be available.
However, as the hospital grew in size and stature, the need for a dedicated research wing became increasingly apparent. Since its inauguration in 1969, the Lady Davis Institute for Medical Research (LDI) has grown into one of Canada’s leading research facilities. In addition to conducting pure research, it encourages researcher-clinicians to pursue projects that have practical applications, taking them from the bedside to the laboratory and back again.
Over the years, the LDI has attracted outstanding investigators with national and international reputations in such specialised areas as cancer therapeutics, molecular oncology, cell and gene therapy, AIDS/HIV, ageing, hypertension and cardiovascular disease, clinical epidemiology, and the psychosocial aspects of the disease.
With a staff of over 400 employees, including more than 200 investigators and at least 290 trainees, the LDI received nearly $32m in grants and produced a total of 829 peer-reviewed publications in 2023.
Success stories
The LDI has made a significant impact with many of its projects, including the following:
BioPortal
The BQC19-JGH Biobank implements hospital-based genomic medicine to improve clinical care, improve the diagnosis of disease, upgrade the efficiency of test ordering, and enhance the prediction of drug responses.

Building on the success of BQC19, the BioPortal was established in 2022 as a bio-banking programme dedicated to conducting research into a range of diseases. Investigators are using the genetic information collected from a diverse population to better understand risk factors and causes of diseases, with the ultimate goal of improving clinical care.
BioPortal currently focuses on improving care for diabetes and will expand to other diseases.
Brain cell research
A team of researchers from across North America, co-led by a principal investigator at the LDI, has discovered that certain brain cells may be inherently vulnerable to mutations that cause high-grade gliomas (highly aggressive and fatal paediatric brain tumours). These findings could help guide the design of clinical trials for new and improved therapies against the leading cause of cancer-related deaths in children and young adults.
The Scleroderma Patient-centred Intervention Network
The Scleroderma Patient-centred Intervention Network (SPIN), led by a senior LDI investigator, is the largest cohort-based trial infrastructure in any rare disease in the world. It maintains a cohort of over 2,300 active participants in 50 centres in seven countries, in collaboration with more than 100 researchers and patients from almost 15 patient organisations. Individuals and organisations involved in SPIN are working on a novel project to develop, adapt and test new and existing programmes to help people with scleroderma cope with their illness and manage their daily lives.
Plasmonic PCR
A team of LDI researchers has developed a rapid plasmonic point-of-care PCR platform to detect pathogens associated with pandemics or antimicrobial resistance. This was the first plasmonic POC prototype built entirely in Montreal. Plasmonic PCR is simple, quick and efficient, with a prototype that has been successfully tested in detecting chlamydia, gonorrhoea, sars-COV2, HIV, HCV and e. coli.
Virtual biopsy development
One of the most powerful and novel technologies in oncology is the development of assays to detect cancer DNA (ctDNA) in the blood. A lab at the LDI is developing a virtual biopsy for precision oncology – i.e., a platform to detect minimal residual cancer in breast cancer patients treated with chemotherapy and surgery.
This test aims to generate personalised ctDNA assays to accurately predict the prognosis of each patient and determine the indication for further anti-cancer therapy. The project is funded by a grant from Genome Québec to support the translation of this platform to the clinic for patients with early breast cancer.
Dr Rosenberg said: “No doubt, a time traveller from 1934 would be dazzled by these and many other of our developments in innovation and research. However, the impetus behind them would be instantly recognisable: To acquire, adapt or develop the best existing practices and technology and to place them in the caring, compassionate hands of our staff. Today and for nearly a century, we have not simply been waiting for tomorrow to arrive; we are making it happen.”
Please note, this article will also appear in the 19th edition of our quarterly publication.