[ad_1]
In a recent review published in the journal Nutrients, researchers investigated the psychobiotic treatment of depression by restoring microbial balance and regulating the microbiome-gut-brain (MGB) axis.
Depression is a global health concern that causes pain, lost productivity, higher healthcare expenditures, and a high risk of suicide. Dysbiosis, a disruption in gut microbiome homeostasis, can affect the gut-brain axis (GBA), resulting in microbial alterations. Psychobiotics, which have favorable effects on the gut barrier, immunological responses, cortisol expression, and the hypothalamic-pituitary-adrenal (HPA) axis, might be used as a supportive treatment for depression, particularly in treatment-recalcitrant depression (TRD) cases.
 The Power of Psychobiotics in Depression: A Modern Approach through the Microbiota–Gut–Brain Axis: A literature Review. Image Credit: T. L. Furrer / Shutterstock
The Power of Psychobiotics in Depression: A Modern Approach through the Microbiota–Gut–Brain Axis: A literature Review. Image Credit: T. L. Furrer / Shutterstock
About the review
In the present review, researchers presented clinical evidence and elucidated the underlying mechanisms of psychobiotic therapies for depression via their effects on gut-brain communication.
Association between the gut-brain axis and depressive disorders
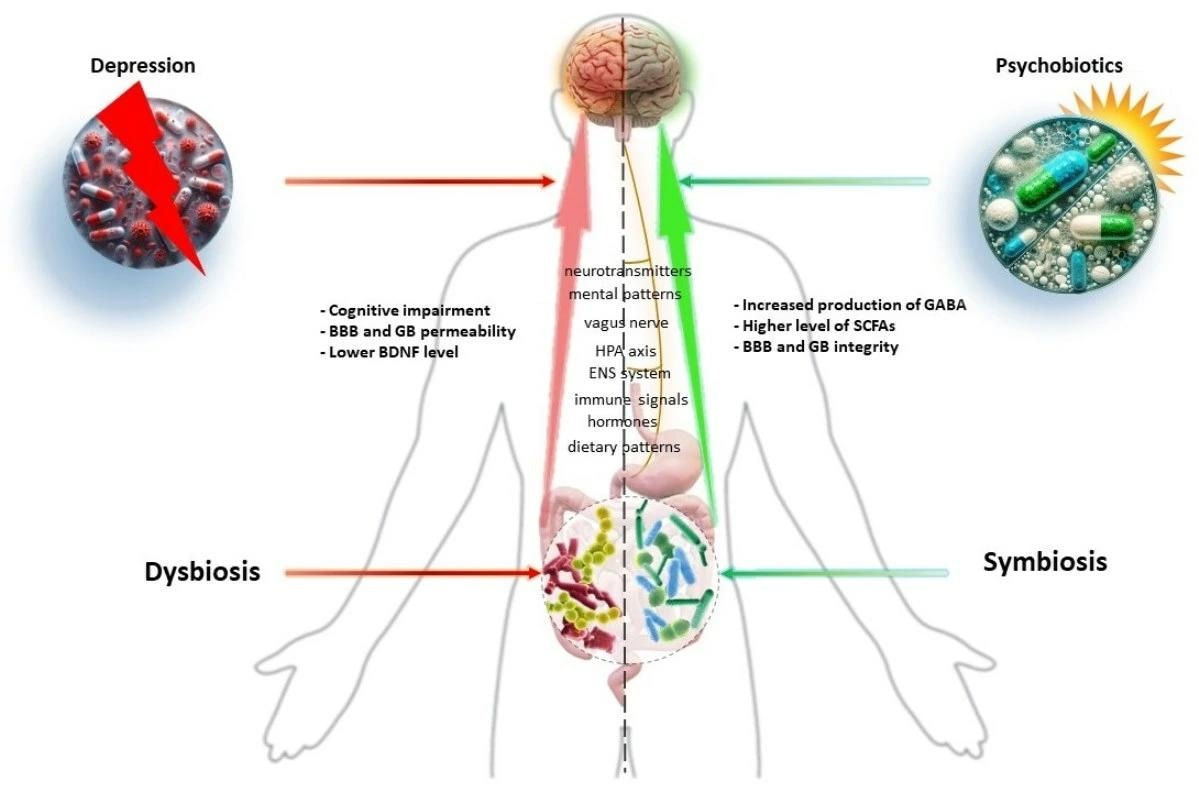
Depression is a complex biological disorder influenced by a variety of molecular mechanisms, such as neurotransmitter reduction, a decrease in brain-derived neurotrophic factor (BDNF), an abnormally stressed HPA axis, an increase in pro-inflammatory gut microbial responses, and vagus nerve interaction between gut microbiota and brain. The GBA and intestinal microbes are inextricably linked, with MGB influencing neurobehavioral outcomes via endocrine, neuronal, and immunological mechanisms. Dysbiosis, or a disruption in the GBA axis, can alter the intestinal microbiome, influencing neuronal function, immunology, and gut inflammation.
Chronic stress impairs intestinal homeostasis and changes gut microbial composition, increasing Faecalibaculum and Clostridium in individuals while decreasing Lactobacillus and Bifidobacterium. Recent animal models have demonstrated a relationship between the gut-brain axis and stress sensitivity and resilience. The intestinal microbiome influences inflammatory responses and brain states and is associated with psychiatric conditions such as major depressive disorder, bipolar disorder, psychosis, schizophrenia, anorexia nervosa, anxiety disorders, obsessive-compulsive disorder, post-traumatic stress disorder, and attention-deficit hyperactivity disorder (ADHD).
Graphical Abstract
Gut microbial metabolites involved in antidepressant actions
The gut microbiome is a vital metabolite source, facilitating communications between the gut and the central nervous system. These metabolites consist of tryptophan, gamma-aminobutyric acid (GABA), serotonin, histamine, 5-hydroxytryptamine (5-HT), short-chain fatty acids (SCFAs), acetylcholine, and dopamine (DA). Microbial metabolites impact various mechanisms important for mental health, such as immunological and neuroendocrine system development, nutrition metabolism modulation, and xenobiotic transformation. They also help to maintain gut barrier function, strengthen the intestinal mucosa, and keep dangerous infections and poisons out of circulation. SCFAs are necessary for emotional states and cognition, impacting the host’s brain via G-protein-coupled receptors. They supply energy to colonocytes, protect the intestinal barrier, regulate inflammatory responses, and regulate hunger hormones. Increased SCFAs can reduce neuroinflammation and boost BDNF synthesis, boosting brain neuroplasticity.
Impact of probiotic gut microbes on depression
Psychobiotics are probiotic bacteria that boost mental health by improving the intestinal barrier and modifying the immune response in the gut-associated lymphoid tissue (GALT), which plays a role in inflammation development. The gut microbiota is crucial in the pathophysiology of depression since it regulates inflammatory processes. Bifidobacterium breve boosts BDNF levels, lowers interleukin-6 (IL-60) and TNF-alpha (TNF-α) levels, and enhances cognitive function.
Lactic acid bacteria (LAB) reduce neuroinflammation, lower kynurenine levels, and promote tight junction (TJ) expression. Lactobacillus plantarum 299v boosts dopamine levels and helps with selective serotonin reuptake inhibitor (SSRI) therapy, resulting in better cognitive performance and lower kynurenine levels. Akkermansia muciniphila suppresses inflammatory cytokines in microglial cells, which lowers depressive-like behavior. Clostridium butyricum protects against neurological dysfunction, whereas Faecalibacterium prausnitzii lowers corticosterone and C-reactive protein (CRP) levels while boosting IL-10 levels and lowering cognitive impairment in Alzheimer’s disease rats.
Clinical evidence highlighting the psychobiotic features of bacterial strains
Postbiotics such as Bacillus coagulans MTCC 5856 and Bifidobacterium longum 1714 can help with irritable bowel syndrome (IBS) symptoms and depression. Probiotics such as Bifidobacterium longum 1714 and NCC3001 help to decrease stress and enhance memory. When coupled with antidepressants, these probiotics can effectively cure TRD. Probiotics such as Lactobacillus casei Shirota and Lactobacillus gasseri CP2305, at 2.5 × 109 CFU/g, enhance general health and lower mood disorders. Multi-strain probiotic medication also boosts general health, alleviates anxiety symptoms, and reduces inflammation. Lactobacillus gasseri fermented black soybean beverage helps healthy individuals sleep better and feel less stressed. Probiotic milk drinks and fermented soybean seed paste improve cognitive performance in individuals with moderate cognitive impairment and Alzheimer’s disease.
The review highlights probiotics’ involvement in lowering depressive symptoms and their importance in mental health. The gut microbiota is crucial for digestion, food absorption, and psychiatric concerns such as stress reduction and anxiety. With a shift in the emphasis in modern life from infectious disorders to more common mental illnesses such as depression, good dietary habits and optimal intestinal function are critical for mental well-being, with probiotics playing an important role.
Journal reference:
- Dziedzic, A.; Maciak, K.; Bliźniewska-Kowalska, K.; Gałecka, M.; Kobierecka, W.; Saluk, J. The Power of Psychobiotics in Depression: A Modern Approach through the Microbiota–Gut–Brain Axis: A literature Review. Nutrients 2024, 16, 1054. DOI: 10.3390/nu16071054, https://www.mdpi.com/2072-6643/16/7/1054
[ad_2]
Source link